Efficacy of immunotherapy in RET fusion-positive NSCLC: A meta-analysis
- PMID: 39149080
- PMCID: PMC11324980
- DOI: 10.1016/j.heliyon.2024.e34626
Efficacy of immunotherapy in RET fusion-positive NSCLC: A meta-analysis
Abstract
Background: The Rearranged during Transfection (RET) gene represents a rare driver mutation in non-small cell lung cancer (NSCLC) occurring in only 1 %-2 % of cases, with implications in targeted carcinogenesis. Despite the significant efficacy demonstrated by immunotherapy in advanced NSCLC with wild-type driver genes, its validation in RET fusion-positive patients is yet to be established.
Objectives: This meta-analysis aims to systematically evaluate the effectiveness of immunotherapy in patients with RET fusion-positive NSCLC.
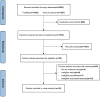
Data sources: and Methods: PubMed and Web of Science databases were systematically searched for relevant studies. Outcomes including objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS), and overall survival (OS) were extracted for further analysis.
Results: Ten real-world evidence (RWE) studies involving 7145 patients were enrolled in this meta-analysis. In terms of tumor response, the pooled ORR and DCR were 24.0 % and 61.0 %, respectively. Regarding survival analysis, the pooled median PFS and median OS were 4.17 months [95 % confidence interval (CI): 3.40-5.02) and 17.22 months (95 % CI: 11.58-23.91)], respectively. Subgroup analyses showed that immunotherapies plus chemotherapy were superior to single-immunotherapy in terms of ORR, DCR, and median PFS, which were 43 % (95 % CI: 31%-55 %) vs. 17 % (95 % CI: 11%-25 %), 74 % (95 % CI: 60%-84 %) vs. 45 % (95 % CI: 31%-59 %) and 6.69 months (95 % CI: 4.91-8.93) vs. 2.96 months (95 % CI: 2.25-3.78), respectively.
Conclusions: To date, RET fusions appear to be associated with poor response to immunotherapy in NSCLC patients, and immunotherapy combined with chemotherapy seems to offer greater clinical benefits than mono-immunotherapy.
Keywords: Immunotherapy; Meta-analysis; Non-small cell lung cancer; RET fusion.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




Similar articles
-
Efficacy of immunotherapy in patients with oncogene-driven non-small-cell lung cancer: a systematic review and meta-analysis.Ther Adv Med Oncol. 2024 Feb 27;16:17588359231225036. doi: 10.1177/17588359231225036. eCollection 2024. Ther Adv Med Oncol. 2024. PMID: 38420602 Free PMC article. Review.
-
The efficacy and safety of selective RET inhibitors in RET fusion-positive non-small cell lung cancer: a meta-analysis.Invest New Drugs. 2023 Oct;41(5):768-776. doi: 10.1007/s10637-023-01390-3. Epub 2023 Aug 21. Invest New Drugs. 2023. PMID: 37603207 Free PMC article. Review.
-
Comparison of the profiles of first-line PD-1/PD-L1 inhibitors for advanced NSCLC lacking driver gene mutations: a systematic review and Bayesian network meta-analysis.Ther Adv Chronic Dis. 2023 Oct 11;14:20406223231189224. doi: 10.1177/20406223231189224. eCollection 2023. Ther Adv Chronic Dis. 2023. PMID: 37841212 Free PMC article.
-
Pralsetinib in RET fusion-positive non-small-cell lung cancer: A real-world data (RWD) analysis from the Italian expanded access program (EAP).Lung Cancer. 2022 Dec;174:118-124. doi: 10.1016/j.lungcan.2022.11.005. Epub 2022 Nov 9. Lung Cancer. 2022. PMID: 36379124
-
The effect of PD-L1/PD-1 immunotherapy in the treatment of squamous non-small-cell lung cancer: a meta-analysis of randomized controlled clinical trials.J Thorac Dis. 2019 Nov;11(11):4453-4463. doi: 10.21037/jtd.2019.11.12. J Thorac Dis. 2019. PMID: 31903233 Free PMC article.
References
LinkOut - more resources
Full Text Sources

