Glutamatergic Modulators for Major Depression from Theory to Clinical Use
- PMID: 39150594
- PMCID: PMC11486832
- DOI: 10.1007/s40263-024-01114-y
Glutamatergic Modulators for Major Depression from Theory to Clinical Use
Abstract
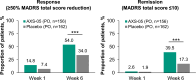
Major depressive disorder (MDD) is a chronic, burdensome, highly prevalent disease that is characterized by depressed mood and anhedonia. MDD is especially burdensome as approved monoamine antidepressant treatments have weeks-long delays before clinical benefit and low remission rates. In the past 2 decades, a promising target emerged to improve patient outcomes in depression treatment: glutamatergic signaling. This narrative review provides a high-level overview of glutamate signaling in synaptogenesis and neural plasticity and the implications of glutamate dysregulation in depression. Based on this preclinical evidence implicating glutamate in depression and the rapid improvement of depression with ketamine treatment in a proof-of-concept trial, a range of N-methyl-D-aspartate (NMDA)-targeted therapies have been investigated. While an array of treatments has been investigated in registered phase 2 or 3 clinical trials, the development of most of these agents has been discontinued. Multiple glutamate-targeted antidepressants are actively in development, and two are approved. Nasal administration of esketamine (Spravato®) was approved by the US Food and Drug Administration (FDA) in 2019 to treat adults with treatment-resistant depression and in 2020 for adults with MDD with acute suicidal ideation or behavior. Oral combination dextromethorphan-bupropion (AXS-05, Auvelity® extended-release tablet) was FDA approved in 2022 for the treatment of MDD in adults. These approvals bolster the importance of glutamate in depression and represent an exciting breakthrough in contemporary psychiatry, providing new avenues of treatment for patients as first-line therapy or with either poor response or unacceptable side effects to monoaminergic antidepressants.
Plain language summary
Major depressive disorder (MDD) is a common disease defined by sadness and a loss of interest or pleasure in activities. Depression treatments include therapy and antidepressant medication. Most available antidepressants affect the same types of chemicals that communicate in the brain (neurotransmitters) called monoamines. Unfortunately, these medicines can take weeks to work for some people and many still have depression symptoms with treatment. To provide more treatment options, new medicines have been studied that impact a different neurotransmitter in the brain, that is, glutamate. Glutamate, the most abundant neurotransmitter, is important for the growth of new brain cell connections, and there are changes in glutamate in people with depression versus people without depression. In 2000, the first small study in people showed that ketamine, a medicine targeting glutamate, quickly improved depression symptoms. Safety risks, including dissociation and sedation, require supervised administration and management guidance. Since 2000, about 20 glutamate-targeted medicines have been tested in human clinical trials. Two of these are approved as antidepressants. The medicine esketamine, which is inhaled up the nose at a clinic under medical supervision, is approved for the adjunctive treatment of people with MDD whose symptoms do not improve with other treatments or who have suicidal thoughts or actions (brand name Spravato®). The combination medicine dextromethorphan–bupropion is a swallowed tablet taken twice daily that is an approved monotherapy to treat adults with MDD (brand name Auvelity®). The approval of these drugs helps show the importance of glutamate in depression and provides more options for people with depression.
© 2024. The Author(s).
Conflict of interest statement
R. Jain received speakers’ fees from Alkermes, Corium, Intra-Cellular Therapies, Ironshore Pharmaceuticals, Lilly, Lundbeck, Merck, Neos Therapeutics, Otsuka, Pamlab, Pfizer, Shire, Sunovion, Takeda, and Tris Pharmaceuticals; consultant fees from AbbVie, Acadia, Adamas, Alfasigma, Cingulate Therapeutics, Eisai, Evidera, Impel, Janssen, Lilly, Lundbeck, Merck, Neos Therapeutics, Neurocrine Biosciences, Osmotica, Otsuka, Pamlab, Pfizer, Shire, Sunovion, Supernus, Takeda, and Teva; is an advisory board member for Adamas, Alkermes, Janssen, Lilly, Lundbeck, Merck, Neos Therapeutics, Neurocrine Biosciences, Otsuka, Pamlab, Pfizer, Shire, Sunovion, Supernus, Takeda, and Teva; and received research support from AbbVie, Lilly, Lundbeck, Otsuka, Pfizer, Shire, and Takeda. R. S. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC); and speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular Therapies, Inc., NewBridge Pharmaceuticals, AbbVie, and Atai Life Sciences. He is CEO of Braxia Scientific Corp (a company focused on ketamine treatment and psychedelic medicine for advanced mental health treatments).
Figures




References
-
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2017;392(10159):1789–858. 10.1016/s0140-6736(18)32279-7. - PMC - PubMed
-
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington: American Psychiatric Publishing; 2013.
-
- Franken IH, Rassin E, Muris P. The assessment of anhedonia in clinical and non-clinical populations: further validation of the Snaith–Hamilton Pleasure Scale (SHAPS). J Affect Disord. 2007;99(1–3):83–9. 10.1016/j.jad.2006.08.020. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

