Sequential high-dose methotrexate and cytarabine administration improves outcomes in real-world patients with primary central nervous system lymphoma: A report from the Australasian Lymphoma Alliance
- PMID: 39157596
- PMCID: PMC11327773
- DOI: 10.1002/jha2.951
Sequential high-dose methotrexate and cytarabine administration improves outcomes in real-world patients with primary central nervous system lymphoma: A report from the Australasian Lymphoma Alliance
Abstract
Background: Despite recent advances, optimal therapeutic approaches applicable to subpopulations with primary central nervous system (CNS) lymphoma outside of clinical trials remain to be determined.
Methods: We performed a retrospective study of immunocompetent, adult patients with histologically confirmed diffuse large B-cell lymphoma of the CNS (PCNSL). 190/204 (93%) patients (median age: 65) received one of five high-dose methotrexate (HD-MTX) containing chemotherapy regimens: MPV/Ara-C (HD-MTX, procarbazine, and vincristine, followed by cytarabine [Ara-C]) (n = 94, 50%), MATRix (HD-MTX, Ara-C, thiotepa, and rituximab) (n = 19, 10%), HD-MTX/Ara-C (n = 31, 16%), HD-MTX monotherapy (n = 35, 18%) and MBVP (HD-MTX, carmustine, teniposide, prednisolone) (n = 11, 6%).
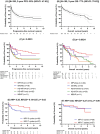
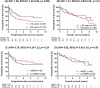
Results: Cumulative median HD-MTX and Ara-C doses were 17 g/m2 (range: 1-64 g/m2) and 12 g/m2 (0-32 g/m2) respectively. Using 14 g/m2 as the reference dose, the median HD-MTX relative dose intensity (HD-MTX-RDI) was 1.25 (0.27-4.57) with 84% receiving > 0.75. The overall response rate (ORR) was 72% (complete response: 50%) after completing HD-MTX. At a median follow-up of 3.41 years (0.06-9.42), progression-free survival (PFS) and overall survival (OS) were different between chemotherapy cohorts, with the best outcomes achieved in the MPV/Ara-C cohort (2-year PFS 74%, 2-year OS 82%; p = 0.0001 and p = 0.0024 respectively). On multivariate analysis, MPV/Ara-C administration and HD-MTX-RDI > 0.75 were associated with longer PFS and OS.
Conclusion: Sequential, response-adapted approaches can improve outcomes, even in older patients who are ineligible for a high-intensity concurrent chemotherapy approach and do not undergo traditional consolidative strategies.
Keywords: DLBCL; PCNSL; cytarabine; high‐dose therapy; methotrexate; retrospective studies.
© 2024 The Author(s). eJHaem published by British Society for Haematology and John Wiley & Sons Ltd.
Conflict of interest statement
Katharine Louise Lewis: Consultancy/Honoraria: Jansen Roche. Trial steering committee: Loxo/Lilly. Advisory: IQVIA and MSD; Pietro R. Di Ciaccio: Honoraria/travel funding: Janssen, Astra Zeneca, and Gilead/Kite; Chan Y. Cheah: Research funding: Beigene. Advisory: Janssen Cilag. Joel Wight: Honoraria: Janssen, MDI, Beigene, MSD, and Abbvie. Travel support: Kite/Gilead. Jake Shortt: Consultancy: BMS, Otsuka, Pfizer, and Astellas. Speaker's Bureau: Mundipharma and Novartis. Research funding: Astex. Dipti Talaulikar: Consultancy/Advisory/Honoraria: Roche, Janssen, Beigene, Novartis, Amgen, Antengene, CSL, EUSA, BMS, and Takeda. Research funding: Roche, Janssen, and Takeda. Tara Cochrane: Research funding: Beigene. Advisory: Janssen Cilag. Eliza A. Hawkes: Consultancy/Advisory: AstraZeneca, Janssen Oncology, Merck Sharpe & Dohme, Gilead Sciences, Bristol Myers Squibb, Novartis, Beigene, Link Healthcare, and Regeneron. Speaker's Bureau: Roche/Genentech, Regeneron, and Abbvie. Research funding: AstraZeneca, Celgene, Merck KGaA, Janssen‐Cilag, Gilead Sciences, Mundipharma, Bristol‐Myers Squibb, and Roche/Genentech. Travel: AstraZeneca.
Figures

References
-
- Swerdlow S, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. 4th ed. Lyon, France: IARC Publications; 2017.
-
- Lippens RJJ, Winograd B. Methotrexate concentration levels in the cerebrospinal fluid during high‐dose methotrexate infusions: an unreliable prediction. Pediatr Hematol Oncol. 1988;5(2):115–124. - PubMed
-
- Martinez‐Calle N, Poynton E, Alchawaf A, Kassam S, Horan M, Rafferty M, et al. Outcomes of older patients with primary central nervous system lymphoma treated in routine clinical practice in the UK: methotrexate dose intensity correlates with response and survival. Br J Haematol. 2020;190(3):394–404. - PubMed
-
- Tatarczuch M, Paul E, Gilberston M, Gregory GP, Tam C, Quach H, et al. Excellent outcomes in older patients with primary CNS lymphoma treated with R‐MPV/cytarabine without whole brain radiotherapy or autologous stem cell transplantation therapy. Leuk Lymphoma. 2021;62(1):112–117. - PubMed
LinkOut - more resources
Full Text Sources
