KASL clinical practice guidelines for noninvasive tests to assess liver fibrosis in chronic liver disease
- PMID: 39159947
- PMCID: PMC11493350
- DOI: 10.3350/cmh.2024.0506
KASL clinical practice guidelines for noninvasive tests to assess liver fibrosis in chronic liver disease
Keywords: Chronic liver disease; Guidelines; Liver fibrosis; Noninvasive test.
Conflict of interest statement
Conflicts of interest statement is available at the official website of Clinical and Molecular Hepatology (Supplementary Table 2,
Figures
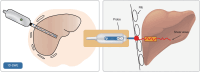
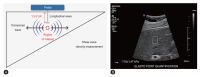
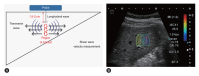
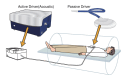
References
-
- Rockey DC, Caldwell SH, Goodman ZD, Nelson RC, Smith AD, American Association for the Study of Liver Diseases Liver biopsy. Hepatology. 2009;49:1017–1044. - PubMed
-
- Poynard T, Munteanu M, Imbert-Bismut F, Charlotte F, Thabut D, Le Calvez S, et al. Prospective analysis of discordant results between biochemical markers and biopsy in patients with chronic hepatitis C. Clin Chem. 2004;50:1344–1355. - PubMed
-
- Rosenberg WM, Voelker M, Thiel R, Becka M, Burt A, Schuppan D, et al. Serum markers detect the presence of liver fibrosis: a cohort study. Gastroenterology. 2004;127:1704–1713. - PubMed
-
- Anstee QM, Castera L, Loomba R. Impact of non-invasive biomarkers on hepatology practice: past, present and future. J Hepatol. 2022;76:1362–1378. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

