Subtyping of gastric cancer based on basement membrane genes that stratifies the prognosis, immune infiltration and therapeutic response
- PMID: 39164593
- PMCID: PMC11336019
- DOI: 10.1007/s12672-024-01238-z
Subtyping of gastric cancer based on basement membrane genes that stratifies the prognosis, immune infiltration and therapeutic response
Abstract
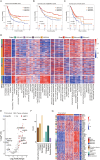
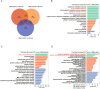
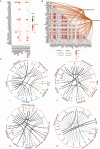
Gastric cancer (GC) is highly heterogeneous and prone to metastasis, which are obstacles to the effectiveness of treatment. The basement membrane (BM) acts as a barrier to tumor cell invasion and metastasis. It is critical to investigate the relationship between BM status, metastasis, and patient prognosis. In several large cohorts, we investigated BM gene expression-based molecular classification and risk-prognosis models for GC, examined tumor microenvironment (TME) differences among different molecular subtypes, and developed risk models in predicting prognosis, immunotherapy effectiveness, and chemotherapy resistance. Three GC subtypes (BMclusterA/B/C) based on BM gene expression status were discovered. Each of the three GC subtypes has unique immune infiltration and activated oncogenic signals. Moreover, a 6-gene score (BMscore) predictive model was developed. The low BMscore group had a high tumor mutation burden, high immunogenicity, and low RHOJ expression levels, implying that individuals with GC in this category may be more susceptible to immunotherapy and treatment. The EMT subtype showed a considerably higher BMscore than the other subtypes in the Asian Organization for Research on Cancer (ACRG) molecular classification. Endothelial cells, smooth muscle cells, and fibroblasts may be engaged in regulating BM reorganization in GC progression, according to single-cell transcriptome analyses. In conclusion, we defined a novel molecular classification of GC based on BM genes, developed a prognostic risk model, and elucidated the cell subpopulations involved in BM remodeling at the single-cell level. This study has deepened the understanding of the relationship between GC metastasis and BM alterations, achieved prognostic stratification, and guided therapy.
Keywords: Basement membrane; Gastric cancer; Metastasis; Molecular classification; Pathology.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Identification of Basement Membrane-Related Signatures in Gastric Cancer.Diagnostics (Basel). 2023 May 25;13(11):1844. doi: 10.3390/diagnostics13111844. Diagnostics (Basel). 2023. PMID: 37296697 Free PMC article.
-
Identification of cuproptosis-related subtypes, construction of a prognosis model, and tumor microenvironment landscape in gastric cancer.Front Immunol. 2022 Nov 21;13:1056932. doi: 10.3389/fimmu.2022.1056932. eCollection 2022. Front Immunol. 2022. PMID: 36479114 Free PMC article.
-
Screening of Differentially Expressed Genes Based on the ACRG Molecular Subtypes of Gastric Cancer and the Significance and Mechanism of AGTR1 Gene Expression.J Pers Med. 2023 Mar 20;13(3):560. doi: 10.3390/jpm13030560. J Pers Med. 2023. PMID: 36983741 Free PMC article.
-
RHOJ Induces Epithelial-to-Mesenchymal Transition by IL-6/STAT3 to Promote Invasion and Metastasis in Gastric Cancer.Int J Biol Sci. 2023 Aug 21;19(14):4411-4426. doi: 10.7150/ijbs.81972. eCollection 2023. Int J Biol Sci. 2023. PMID: 37781036 Free PMC article.
-
Characterization of the Prognosis and Tumor Microenvironment of Cellular Senescence-related Genes through scRNA-seq and Bulk RNA-seq Analysis in GC.Recent Pat Anticancer Drug Discov. 2024;19(4):530-542. doi: 10.2174/0115748928255417230924191157. Recent Pat Anticancer Drug Discov. 2024. PMID: 37807645
References
-
- Yang L, Kartsonaki C, Yao P, de Martel C, Plummer M, Chapman D, et al. The relative and attributable risks of cardia and non-cardia gastric cancer associated with Helicobacter pylori infection in China: a case-cohort study. Lancet Public Health. 2021;6(12):e888–96. 10.1016/S2468-2667(21)00164-X - DOI - PMC - PubMed
-
- Shitara K, Van Cutsem E, Bang YJ, Fuchs C, Wyrwicz L, Lee KW, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial. JAMA Oncol. 2020;6(10):1571–80. 10.1001/jamaoncol.2020.3370 - DOI - PMC - PubMed
Grants and funding
- YJKJJ06/Pharmaceutical Research and Exploration Fund
- 2022048/the Hefei Municipal Natural Science Foundation
- 2022050/the Hefei Municipal Natural Science Foundation
- 81872276/the National Natural Science Foundation of China
- LMPT201908/the foundation of Anhui Province Key Laboratory of Medical Physics and Technology
LinkOut - more resources
Full Text Sources
Miscellaneous
