Advanced glycation end products mediate biomineralization disorder in diabetic bone disease
- PMID: 39173634
- PMCID: PMC11524989
- DOI: 10.1016/j.xcrm.2024.101694
Advanced glycation end products mediate biomineralization disorder in diabetic bone disease
Abstract
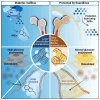
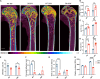
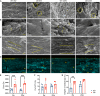
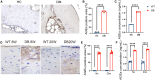
Patients with diabetes often experience fragile fractures despite normal or higher bone mineral density (BMD), a phenomenon termed the diabetic bone paradox (DBP). The pathogenesis and therapeutics opinions for diabetic bone disease (DBD) are not fully explored. In this study, we utilize two preclinical diabetic models, the leptin receptor-deficient db/db mice (DB) mouse model and the streptozotocin-induced diabetes (STZ) mouse model. These models demonstrate higher BMD and lower mechanical strength, mirroring clinical observations in diabetic patients. Advanced glycation end products (AGEs) accumulate in diabetic bones, causing higher non-enzymatic crosslinking within collagen fibrils. This inhibits intrafibrillar mineralization and leads to disordered mineral deposition on collagen fibrils, ultimately reducing bone strength. Guanidines, inhibiting AGE formation, significantly improve the microstructure and biomechanical strength of diabetic bone and enhance bone fracture healing. Therefore, targeting AGEs may offer a strategy to regulate bone mineralization and microstructure, potentially preventing the onset of DBD.
Keywords: AGEs; advanced glycation end products; aminoguandine; biomineralization; bone fracture; bone quality; collagen mineralization; diabetes bone disease; metformin; type 2 diabetes mellitus.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures



Similar articles
-
Advanced Glycation End Products, Diabetes, and Bone Strength.Curr Osteoporos Rep. 2016 Dec;14(6):320-326. doi: 10.1007/s11914-016-0332-1. Curr Osteoporos Rep. 2016. PMID: 27704396 Free PMC article. Review.
-
Induction and rescue of skeletal fragility in a high-fat diet mouse model of type 2 diabetes: An in vivo and in vitro approach.Bone. 2022 Mar;156:116302. doi: 10.1016/j.bone.2021.116302. Epub 2021 Dec 21. Bone. 2022. PMID: 34952229 Free PMC article.
-
Decoding the diabetic bone paradox: How AGEs sabotage skeletal integrity.Cell Rep Med. 2024 Sep 17;5(9):101693. doi: 10.1016/j.xcrm.2024.101693. Cell Rep Med. 2024. PMID: 39293394 Free PMC article.
-
Treatment With Small Molecule Inhibitors of Advanced Glycation End-Products Formation and Advanced Glycation End-Products-Mediated Collagen Cross-Linking Promotes Experimental Aortic Aneurysm Progression in Diabetic Mice.J Am Heart Assoc. 2023 May 16;12(10):e028081. doi: 10.1161/JAHA.122.028081. Epub 2023 May 9. J Am Heart Assoc. 2023. PMID: 37158066 Free PMC article.
-
Effects of Diabetes on Bone Material Properties.Curr Osteoporos Rep. 2019 Dec;17(6):455-464. doi: 10.1007/s11914-019-00538-6. Curr Osteoporos Rep. 2019. PMID: 31713179 Free PMC article. Review.
Cited by
-
Bidirectional modulation of glycolysis using a multifunctional nanocomposite hydrogel promotes bone fracture healing in type 2 diabetes mellitus.Bioact Mater. 2025 Apr 8;50:152-170. doi: 10.1016/j.bioactmat.2025.03.020. eCollection 2025 Aug. Bioact Mater. 2025. PMID: 40256330 Free PMC article.
-
Localized sclerostin accumulation in osteocyte lacunar-canalicular system is associated with cortical bone microstructural alterations and bone fragility in db/db male mice.Front Cell Dev Biol. 2025 Apr 25;13:1562764. doi: 10.3389/fcell.2025.1562764. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40352664 Free PMC article.
-
Influence of Overweight and Obesity on Bone Remodeling during Pregnancy.Res Sq [Preprint]. 2025 Jun 2:rs.3.rs-6606324. doi: 10.21203/rs.3.rs-6606324/v1. Res Sq. 2025. PMID: 40502759 Free PMC article. Preprint.
-
Ageing-Related Changes in Ultrastructural Bone Matrix Composition and Osteocyte Mechanosensitivity.Curr Osteoporos Rep. 2025 Aug 12;23(1):35. doi: 10.1007/s11914-025-00927-0. Curr Osteoporos Rep. 2025. PMID: 40794153 Free PMC article. Review.
-
Multifunctional hydrogel loaded with 4-octyl itaconate and exosomes to induce bone regeneration for diabetic infected bone defect via Keap1-Nrf2 pathway.Mater Today Bio. 2025 Feb 19;31:101588. doi: 10.1016/j.mtbio.2025.101588. eCollection 2025 Apr. Mater Today Bio. 2025. PMID: 40070866 Free PMC article.
References
-
- Bonds D.E., Larson J.C., Schwartz A.V., Strotmeyer E.S., Robbins J., Rodriguez B.L., Johnson K.C., Margolis K.L. Risk of fracture in women with type 2 diabetes: the Women’s Health Initiative Observational Study. J. Clin. Endocrinol. Metab. 2006;91:3404–3410. - PubMed
-
- Nethander M., Coward E., Reimann E., Grahnemo L., Gabrielsen M.E., Wibom C., Estonian Biobank Research Team. Mägi R., Funck-Brentano T., Hoff M., et al. Assessment of the genetic and clinical determinants of hip fracture risk: Genome-wide association and Mendelian randomization study. CR Med. 2022;3 doi: 10.1016/j.xcrm.2022.100776. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

