Integrating molecular and cellular components of endothelial shear stress mechanotransduction
- PMID: 39178024
- PMCID: PMC11482243
- DOI: 10.1152/ajpheart.00431.2024
Integrating molecular and cellular components of endothelial shear stress mechanotransduction
Abstract
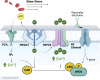
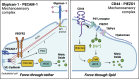
The lining of blood vessels is constantly exposed to mechanical forces exerted by blood flow against the endothelium. Endothelial cells detect these tangential forces (i.e., shear stress), initiating a host of intracellular signaling cascades that regulate vascular physiology. Thus, vascular health is tethered to the endothelial cells' capacity to transduce shear stress. Indeed, the mechanotransduction of shear stress underlies a variety of cardiovascular benefits, including some of those associated with increased physical activity. However, endothelial mechanotransduction is impaired in aging and disease states such as obesity and type 2 diabetes, precipitating the development of vascular disease. Understanding endothelial mechanotransduction of shear stress, and the molecular and cellular mechanisms by which this process becomes defective, is critical for the identification and development of novel therapeutic targets against cardiovascular disease. In this review, we detail the primary mechanosensitive structures that have been implicated in detecting shear stress, including junctional proteins such as platelet endothelial cell adhesion molecule-1 (PECAM-1), the extracellular glycocalyx and its components, and ion channels such as piezo1. We delineate which molecules are truly mechanosensitive and which may simply be indispensable for the downstream transmission of force. Furthermore, we discuss how these mechanosensors interact with other cellular structures, such as the cytoskeleton and membrane lipid rafts, which are implicated in translating shear forces to biochemical signals. Based on findings to date, we also seek to integrate these cellular and molecular mechanisms with a view of deciphering endothelial mechanotransduction of shear stress, a tenet of vascular physiology.
Keywords: blood flow; cytoskeleton; endothelium; glycocalyx; mechanosensation.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

