Antifungal potential, mechanism of action, and toxicity of 1,4-naphthoquinone derivatives
- PMID: 39178045
- PMCID: PMC11393642
- DOI: 10.1556/1886.2024.00072
Antifungal potential, mechanism of action, and toxicity of 1,4-naphthoquinone derivatives
Abstract
Background: The rising prevalence of fungal infections and challenges such as adverse effects and resistance against existing antifungal agents have driven the exploration of new antifungal substances.
Methods: We specifically investigated naphthoquinones, known for their broad biological activities and promising antifungal capabilities. It specifically examined the effects of a particular naphthoquinone on the cellular components of Candida albicans ATCC 60193. The study also assessed cytotoxicity in MRC-5 cells, Artemia salina, and the seeds of tomatoes and arugula.
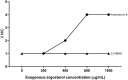
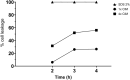
Results: Among four tested naphthoquinones, 2,3-DBNQ (2,3-dibromonaphthalene-1,4-dione) was identified as highly effective, showing potent antifungal activity at concentrations between 1.56 and 6.25 μg mL-1. However, its cytotoxicity in MRC-5 cells (IC50 = 15.44 µM), complete mortality in A. salina at 50 μg mL-1, and significant seed germination inhibition suggest limitations for its clinical use.
Conclusions: The findings indicate that primary antifungal mechanism of 2,3-DBNQ might involve disrupting fungal membrane permeability, which leads to increased nucleotide leakage. This insight underscores the need for further research to enhance the selectivity and safety of naphthoquinones for potential therapeutic applications.
Keywords: 2,3-DBNQ; antifungal potential; cytotoxicity; fungal membrane permeability; naphthoquinone derivatives.
Conflict of interest statement
Figures

Similar articles
-
1,4-Naphthoquinone derivatives potently suppress Candida albicans growth, inhibit formation of hyphae and show no toxicity toward zebrafish embryos.J Med Microbiol. 2018 Apr;67(4):598-609. doi: 10.1099/jmm.0.000700. Epub 2018 Feb 20. J Med Microbiol. 2018. PMID: 29461185
-
Essential Oil of Cymbopogon nardus (L.) Rendle: A Strategy to Combat Fungal Infections Caused by Candida Species.Int J Mol Sci. 2016 Aug 9;17(8):1252. doi: 10.3390/ijms17081252. Int J Mol Sci. 2016. PMID: 27517903 Free PMC article.
-
The Antifungal Peptide MCh-AMP1 Derived From Matricaria chamomilla Inhibits Candida albicans Growth via Inducing ROS Generation and Altering Fungal Cell Membrane Permeability.Front Microbiol. 2020 Jan 21;10:3150. doi: 10.3389/fmicb.2019.03150. eCollection 2019. Front Microbiol. 2020. PMID: 32038583 Free PMC article.
-
New and Promising Chemotherapeutics for Emerging Infections Involving Drug-resistant Non-albicans Candida Species.Curr Top Med Chem. 2019;19(28):2527-2553. doi: 10.2174/1568026619666191025152412. Curr Top Med Chem. 2019. PMID: 31654512 Review.
-
The in vivo anti-Candida albicans activity of flavonoids.J Oral Biosci. 2021 Jun;63(2):120-128. doi: 10.1016/j.job.2021.03.004. Epub 2021 Apr 9. J Oral Biosci. 2021. PMID: 33839266 Review.
Cited by
-
Antifungal Activity of Selected Naphthoquinones and Their Synergistic Combination with Amphotericin B Against Cryptococcus neoformans H99.Antibiotics (Basel). 2025 Jun 13;14(6):602. doi: 10.3390/antibiotics14060602. Antibiotics (Basel). 2025. PMID: 40558192 Free PMC article.
References
-
- Ozturk I, Tunçel A, Yurt F, Biyiklioglu Z, Ince M, Ocakoglu K. Antifungal photodynamic activities of phthalocyanine derivatives on Candida albicans. Photodiagnosis Photodyn Ther. 2020;30:101715. - PubMed
-
- Hendrickson JA, Hu C, Aitken SL, Beyda N. Antifungal resistance: a concerning trend for the present and future. Curr Infect Dis Rep. 2019;21:1–8. - PubMed
-
- Harris E. CDC: Candida auris fungal infections and drug resistance on the rise. JAMA. 2023;329(15):1248–1248. - PubMed
-
- Chowdhary A, Prakash A, Sharma C, Kordalewska M, Kumar A, Sarma S, et al. . A multicentre study of antifungal susceptibility patterns among 350 Candida auris isolates (2009–17) in India: role of the ERG11 and FKS1 genes in azole and echinocandin resistance. J Antimicrob Chemother. 2018;73(4):891–899. - PubMed
-
- Forsberg K, Woodworth K, Walters M, Berkow EL, Jackson B, Chiller T, et al. . Candida auris: the recent emergence of a multidrug-resistant fungal pathogen. Med Myco. 2019;57(1):1–12. - PubMed
LinkOut - more resources
Full Text Sources
