Tau PET positivity predicts clinically relevant cognitive decline driven by Alzheimer's disease compared to comorbid cases; proof of concept in the ADNI study
- PMID: 39179903
- PMCID: PMC11746147
- DOI: 10.1038/s41380-024-02672-9
Tau PET positivity predicts clinically relevant cognitive decline driven by Alzheimer's disease compared to comorbid cases; proof of concept in the ADNI study
Abstract
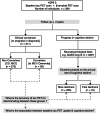
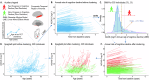
β-amyloid (Aβ) pathology is not always coupled with Alzheimer's disease (AD) relevant cognitive decline. We assessed the accuracy of tau PET to identify Aβ(+) individuals who show prospective disease progression. 396 cognitively unimpaired and impaired individuals with baseline Aβ and tau PET and a follow-up of ≥ 2 years were selected from the Alzheimer's Disease Neuroimaging Initiative dataset. The participants were dichotomously grouped based on either clinical conversion (i.e., change of diagnosis) or cognitive deterioration (fast (FDs) vs. slow decliners (SDs)) using data-driven clustering of the individual annual rates of cognitive decline. To assess cognitive decline in individuals with isolated Aβ(+) or absence of both Aβ and tau (T) pathologies, we investigated the prevalence of non-AD comorbidities and FDG PET hypometabolism patterns suggestive of AD. Baseline tau PET uptake was higher in Aβ(+)FDs than in Aβ(-)FD/SDs and Aβ(+)SDs, independently of baseline cognitive status. Baseline tau PET uptake identified MCI Aβ(+) Converters and Aβ(+)FDs with an area under the curve of 0.85 and 0.87 (composite temporal region of interest) respectively, and was linearly related to the annual rate of cognitive decline in Aβ(+) individuals. The T(+) individuals constituted largely a subgroup of those being Aβ(+) and those clustered as FDs. The most common biomarker profiles in FDs (n = 70) were Aβ(+)T(+) (n = 34, 49%) and Aβ(+)T(-) (n = 19, 27%). Baseline Aβ load was higher in Aβ(+)T(+)FDs (M = 83.03 ± 31.42CL) than in Aβ(+)T(-)FDs (M = 63.67 ± 26.75CL) (p-value = 0.038). Depression diagnosis was more prevalent in Aβ(+)T(-)FDs compared to Aβ(+)T(+)FDs (47% vs. 15%, p-value = 0.021), as were FDG PET hypometabolism pattern not suggestive of AD (86% vs. 50%, p-value = 0.039). Our findings suggest that high tau PET uptake is coupled with both Aβ pathology and accelerated cognitive decline. In cases of isolated Aβ(+), cognitive decline may be associated with changes within the AD spectrum in a multi-morbidity context, i.e., mixed AD.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Consortia: Data used in preparation of this article were obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database ( adni.loni.usc.edu ). As such, the investigators within the ADNI contributed to the design and implementation of ADNI and/or provided data but did not participate in analysis or writing of this report. A complete listing of ADNI investigators can be found at: https://adni.loni.usc.edu/wp-content/uploads/how_to_apply/ADNI_Acknowledgement_List.pdf .
Figures



References
-
- Kadir A, Marutle A, Gonzalez D, Schöll M, Almkvist O, Mousavi M, et al. Positron emission tomography imaging and clinical progression in relation to molecular pathology in the first Pittsburgh Compound B positron emission tomography patient with Alzheimer’s disease. Brain. 2011;134:301–17. - PMC - PubMed
-
- Rowe CC, Bourgeat P, Ellis KA, Brown B, Lim YY, Mulligan R, et al. Predicting Alzheimer disease with β-amyloid imaging: results from the Australian imaging, biomarkers, and lifestyle study of ageing. Ann Neurol. 2013;74:905–13.
-
- Sabri O, Sabbagh MN, Seibyl J, Barthel H, Akatsu H, Ouchi Y, et al. Florbetaben PET imaging to detect amyloid beta plaques in Alzheimer’s disease: Phase 3 study. Alzheimer’s & Dementia. 2015;11:964–74. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

