The Protective Role of Troxerutin (Trox) in Counteracting Anaplastic Thyroid Carcinoma (ATC) Progression
- PMID: 39200219
- PMCID: PMC11351865
- DOI: 10.3390/biomedicines12081755
The Protective Role of Troxerutin (Trox) in Counteracting Anaplastic Thyroid Carcinoma (ATC) Progression
Abstract
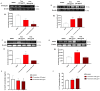
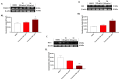
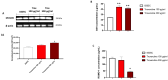
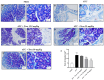
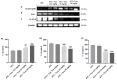
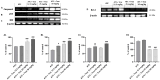
Anaplastic thyroid carcinoma (ATC) is a rare thyroid neoplasm characterized by aggressiveness and a high mortality rate. Troxerutin (Trox) is a bioflavonoid widely found in various fruits and vegetables with numerous protective effects, including anticancer activities. To evaluate the anti-oxidant and anti-inflammatory effect of Trox, in vitro and in vivo studies were conducted in a model of ATC. Human ATC 8305C cell lines were treated with increasing concentrations of Trox (10 μg/mL, 30 μg/mL, 100 μg/mL, 300 μg/mL), and our results revealed that Trox treatment was able to reduce the viability of ATC cells and migratory capacity, reducing the expression of anti-apoptotic factors, such as B-cell lymphoma (bcl-2), and increasing the expression of pro-apoptotic factors, such as Caspase-3 and BID, activating oxidative stress mediators, such as manganese superoxide dismutase (MnSOD), heme oxygenase-1 (HO-1), glutathione (GSH) and reactive oxygen species modulator 1 (ROMO-1). Furthermore, Trox modulates NF-κB pathway markers, such as NIK and TRAF-6. Further confirmation was obtained through in vivo studies, in which Trox treatment, at doses of 12.5, 25 and 50 mg/kg, reduced morphological alteration, decreasing mast cell accumulation. Therefore, the use of Trox could be considered a promising strategy to counteract the progression of ATC.
Keywords: anaplastic thyroid carcinoma (ATC); anti-oxidant activity; antitumoral activity; chemotherapy; troxerutin.
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures






Similar articles
-
Antiangiogenic Potential of Troxerutin and Chitosan Loaded Troxerutin on Chorioallantoic Membrane Model.Biomed Res Int. 2023 May 23;2023:5956154. doi: 10.1155/2023/5956154. eCollection 2023. Biomed Res Int. 2023. PMID: 37260851 Free PMC article.
-
The EPRS-ATF4-COLI pathway axis is a potential target for anaplastic thyroid carcinoma therapy.Phytomedicine. 2024 Jul;129:155670. doi: 10.1016/j.phymed.2024.155670. Epub 2024 Apr 28. Phytomedicine. 2024. PMID: 38704915
-
Inhibition of Cancer Stem-Like Phenotype by Curcumin and Deguelin in CAL-62 Anaplastic Thyroid Cancer Cells.Anticancer Agents Med Chem. 2019;19(15):1887-1898. doi: 10.2174/1871520619666191004144025. Anticancer Agents Med Chem. 2019. PMID: 31584382
-
Alantolactone induces concurrent apoptosis and GSDME-dependent pyroptosis of anaplastic thyroid cancer through ROS mitochondria-dependent caspase pathway.Phytomedicine. 2023 Jan;108:154528. doi: 10.1016/j.phymed.2022.154528. Epub 2022 Oct 27. Phytomedicine. 2023. PMID: 36343549
-
Anaplastic thyroid carcinoma: from clinicopathology to genetics and advanced therapies.Nat Rev Endocrinol. 2017 Nov;13(11):644-660. doi: 10.1038/nrendo.2017.76. Epub 2017 Jul 14. Nat Rev Endocrinol. 2017. PMID: 28707679 Review.
Cited by
-
Casein Kinase 2 Inhibitor, CX-4945, Induces Apoptosis and Restores Blood-Brain Barrier Homeostasis in In Vitro and In Vivo Models of Glioblastoma.Cancers (Basel). 2024 Nov 24;16(23):3936. doi: 10.3390/cancers16233936. Cancers (Basel). 2024. PMID: 39682125 Free PMC article.
References
-
- Haugen B.R., Alexander E.K., Bible K.C., Doherty G.M., Mandel S.J., Nikiforov Y.E., Pacini F., Randolph G.W., Sawka A.M., Schlumberger M., et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26:1–133. doi: 10.1089/thy.2015.0020. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

