Glutathione-S-Transferase Theta 2 (GSTT2) Modulates the Response to Bacillus Calmette-Guérin Immunotherapy in Bladder Cancer Patients
- PMID: 39201633
- PMCID: PMC11354831
- DOI: 10.3390/ijms25168947
Glutathione-S-Transferase Theta 2 (GSTT2) Modulates the Response to Bacillus Calmette-Guérin Immunotherapy in Bladder Cancer Patients
Abstract
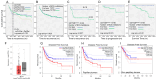
Glutathione-S-transferases (GST) enzymes detoxify xenobiotics and are implicated in response to anticancer therapy. This study evaluated the association of GST theta 1 (GSTT1), GSTT2, and GSTT2B with Mycobacterium bovis Bacillus Calmette-Guérin (BCG) response in non-muscle-invasive bladder cancer treatment. In vitro assessments of GSTT2 knockout (KO) effects were performed using cell lines and dendritic cells (DCs) from GSTT2KO mice. Deletion of GSTT2B, GSTT1, and single-nucleotide polymorphisms in the promoter region of GSTT2 was analysed in patients (n = 205) and healthy controls (n = 150). Silencing GSTT2 expression in MGH cells (GSTT2BFL/FL) resulted in increased BCG survival (p < 0.05) and decreased cellular reactive oxygen species. In our population, there are 24.2% with GSTT2BDel/Del and 24.5% with GSTT2BFL/FL. With ≤ 8 instillations of BCG therapy (n = 51), 12.5% of GSTT2BDel/Del and 53.8% of GSTT2BFL/FL patients had a recurrence (p = 0.041). With ≥9 instillations (n = 153), the disease recurred in 45.5% of GSTT2BDel/Del and 50% of GSTT2BFL/FL. GSTT2FL/FL patients had an increased likelihood of recurrence post-BCG therapy (HR 5.5 [1.87-16.69] p < 0.002). DCs from GSTT2KO mice produced three-fold more IL6 than wild-type DCs, indicating a robust inflammatory response. To summarise, GSTT2BDel/Del patients respond better to less BCG therapy and could be candidates for a reduced surveillance regimen.
Keywords: BCG vaccine; bladder cancer; genetic; glutathione S-transferase theta 2; immunotherapy; polymorphism; urinary bladder neoplasms.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures


References
-
- Babjuk M., Burger M., Capoun O., Cohen D., Compérat E.M., Dominguez Escrig J.L., Gontero P., Liedberg F., Masson-Lecomte A., Mostafid A.H., et al. European Association of Urology Guidelines on Non-muscle-invasive Bladder Cancer (Ta, T1, and Carcinoma in Situ) Eur. Urol. 2022;81:75–94. doi: 10.1016/j.eururo.2021.08.010. - DOI - PubMed
-
- Cambier S., Sylvester R.J., Collette L., Gontero P., Brausi M.A., van Andel G., Kirkels W.J., Silva F.C., Oosterlinck W., Prescott S., et al. EORTC Nomograms and Risk Groups for Predicting Recurrence, Progression, and Disease-specific and Overall Survival in Non-Muscle-invasive Stage Ta-T1 Urothelial Bladder Cancer Patients Treated with 1–3 Years of Maintenance Bacillus Calmette-Guérin. Eur. Urol. 2016;69:60–69. doi: 10.1016/j.eururo.2015.06.045. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

