Regeneration and Plasticity Induced by Epidural Stimulation in a Rodent Model of Spinal Cord Injury
- PMID: 39201729
- PMCID: PMC11354918
- DOI: 10.3390/ijms25169043
Regeneration and Plasticity Induced by Epidural Stimulation in a Rodent Model of Spinal Cord Injury
Abstract
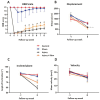
Traumatic spinal cord injury is a major cause of disability for which there are currently no fully effective treatments. Recent studies using epidural electrical stimulation have shown significant advances in motor rehabilitation, even when applied during chronic phases of the disease. The present study aimed to investigate the effectiveness of epidural electric stimulation in the motor recovery of rats with spinal cord injury. Furthermore, we aimed to elucidate the neurophysiological mechanisms underlying motor recovery. First, we improved upon the impact spinal cord injury model to cause severe and permanent motor deficits lasting up to 2 months. Next, we developed and tested an implantable epidural spinal cord stimulator device for rats containing an electrode and an implantable generator. Finally, we evaluated the efficacy of epidural electrical stimulation on motor recovery after spinal cord injury in Wistar rats. A total of 60 animals were divided into the following groups: (i) severe injury with epidural electrical stimulation (injury + stim, n = 15), (ii) severe injury without stimulation (group injury, n = 15), (iii) sham implantation without battery (sham, n = 15), and (iv) a control group, without surgical intervention (control, n = 15). All animals underwent weekly evaluations using the Basso, Beattie, Bresnahan (BBB) locomotor rating scale index, inclined plane, and OpenField test starting one week before the lesion and continuing for eight weeks. After this period, the animals were sacrificed and their spinal cords were explanted and prepared for histological analysis (hematoxylin-eosin) and immunohistochemistry for NeuN, β-III-tubulin, synaptophysin, and Caspase 3. Finally, NeuN-positive neuronal nuclei were quantified through stereology; fluorescence signal intensities for β-tubulin, synaptophyin, and Caspase 3 were quantified using an epifluorescence microscope. The injury + stim group showed significant improvement on the BBB scale compared with the injured group after the 5th week (p < 0.05). Stereological analysis showed a significantly higher average count of neural cells in the injury + stim group in relation to the injury group (1783 ± 2 vs. 897 ± 3, p < 0.001). Additionally, fluorescence signal intensity for synaptophysin was significantly higher in the injury + stim group in relation to the injury group (1294 ± 46 vs. 1198 ± 23, p < 0.01); no statistically significant difference was found in β-III-tubulin signal intensity. Finally, Caspase 3 signal intensity was significantly lower in the stim group (727 ± 123) compared with the injury group (1225 ± 87 p < 0.05), approaching levels observed in the sham and control groups. Our data suggest a regenerative and protective effect of epidural electrical stimulation in rats subjected to impact-induced traumatic spinal cord injury.
Keywords: epidural electric stimulation; motor recovery; neuroplasticity; regeneration; spinal cord injury.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Mariano E.D., Batista C.M., Barbosa B.J., Marie S.K., Teixeira M.J., Morgalla M., Tatagiba M., Li J., Lepski G. Current perspectives in stem cell therapy for spinal cord repair in humans: A review of work from the past 10 years. Arq. Neuropsiquiatr. 2014;72:451–456. doi: 10.1590/0004-282X20140051. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

