Developing an Optimized Protocol for Regeneration and Transformation in Pepper
- PMID: 39202378
- PMCID: PMC11353661
- DOI: 10.3390/genes15081018
Developing an Optimized Protocol for Regeneration and Transformation in Pepper
Abstract
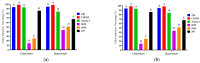
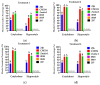
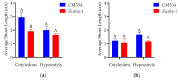
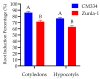
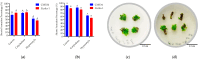
Capsicum annuum L. is extensively cultivated in subtropical and temperate regions globally, respectively, when grown in a medium with 8 holding significant economic importance. Despite the availability of genome sequences and editing tools, gene editing in peppers is limited by the lack of a stable regeneration and transformation method. This study assessed regeneration and transformation protocols in seven chili pepper varieties, including CM334, Zunla-1, Zhongjiao6 (ZJ6), 0818, 0819, 297, and 348, in order to enhance genetic improvement efforts. Several explants, media compositions, and hormonal combinations were systematically evaluated to optimize the in vitro regeneration process across different chili pepper varieties. The optimal concentrations for shoot formation, shoot elongation, and rooting in regeneration experiments were determined as 5 mg/L of 6-Benzylaminopurine (BAP) with 5 mg/L of silver nitrate (AgNO3), 0.5 mg/L of Gibberellic acid (GA3), and 1 mg/L of Indole-3-butyric acid (IBA), respectively. The highest regeneration rate of 41% was observed from CM334 cotyledon explants. Transformation optimization established 300 mg/L of cefotaxime for bacterial control, with a 72-h co-cultivation period at OD600 = 0.1. This study optimizes the protocols for chili pepper regeneration and transformation, thereby contributing to genetic improvement efforts.
Keywords: Agrobacterium tumefaciens; Capsicum annuum; explants; in vitro culture.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures








References
-
- Sharma V.K., Srivastava A., Mangal M. Recent Trends in Sweet Pepper Breeding. In: Gosal S., Wani S., editors. Accelerated Plant Breeding. Volume 2. Springer; Berlin/Heidelberg, Germany: 2020. pp. 417–444.
-
- Pérez-Grajales M., Martínez-Damián M.T., Cruz-Alvarez O., Potrero-Andrade S.M., Peña-Lomelí A., González-Hernández V.A., Villegas-Monter A. Content of capsaicinoids and physicochemical characteristics of manzano hot pepper grown in greenhouse. Not. Bot. Horti Agrobot. 2019;47:119–127. doi: 10.15835/nbha47111241. - DOI
-
- Agarwal A., Gupta S., Ahmed Z. Influence of Plant Densities on Productivity of Bell Pepper (Capsicum annuum L.) under Greenhouse in High Altitude Cold Desert of Ladakh; Proceedings of the International Symposium on Medicinal and Nutraceutical Plants 756; Macon, GA, USA. 8 November 2007; pp. 309–314.
-
- Pawar S.S., Bharude N.V., Sonone S.S., Deshmukh R.S., Raut A.K., Umarkar A.R. Chillies as Food, Spice and Medicine: A Perspective. Int. J. Pharm. Biol. Sci. 2011;1:311–318.
MeSH terms
Substances
Grants and funding
- 2020GBJ002233/China Scholarship Council
- CARS-23-A15/China Agriculture Research System
- CAAS-ASTIP-IVFCAAS/Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences
- 2023YFD1200101/National Key Research and Development program of China
- 32302557/National Natural Science Foundation of China for Youth Scholar
LinkOut - more resources
Full Text Sources

