Immune Checkpoint Inhibitors for Pediatric Cancers: Is It Still a Stalemate?
- PMID: 39204096
- PMCID: PMC11357301
- DOI: 10.3390/ph17080991
Immune Checkpoint Inhibitors for Pediatric Cancers: Is It Still a Stalemate?
Abstract
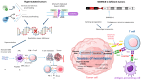
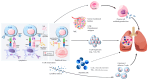
The knowledge surrounding the application of immune checkpoint inhibitors (ICIs) in the treatment of pediatric cancers is continuously expanding and evolving. These therapies work by enhancing the body's natural immune response against tumors, which may have been suppressed by certain pathways. The effectiveness of ICIs in treating adult cancers has been widely acknowledged. However, the results of early phase I/II clinical trials that exclusively targeted the use of ICIs for treating different pediatric cancers have been underwhelming. The response rates to ICIs have generally been modest, except for cases of pediatric classic Hodgkin lymphoma. There seems to be a notable disparity in the immunogenicity of childhood cancers compared to adult cancers, potentially accounting for this phenomenon. On average, childhood cancers tend to have significantly fewer neoantigens. In recent times, there has been a renewed sense of optimism regarding the potential benefits of ICI therapies for specific groups of children with cancer. In initial research, individuals diagnosed with pediatric hypermutated and SMARCB1-deficient cancers have shown remarkable positive outcomes when treated with ICI therapies. This is likely due to the underlying biological factors that promote the expression of neoantigens and inflammation within the tumor. Ongoing trials are diligently assessing the effectiveness of ICIs for pediatric cancer patients in these specific subsets. This review aimed to analyze the safety and effectiveness of ICIs in pediatric patients with different types of highly advanced malignancies.
Keywords: SMARCB1-deficient; hypermutated; immune checkpoint inhibitors; pediatric cancer.
Conflict of interest statement
No conflicts of interest are declared by the authors.
Figures



Similar articles
-
Checkpoint Immunotherapy in Pediatrics: Here, Gone, and Back Again.Am Soc Clin Oncol Educ Book. 2022 Apr;42:1-14. doi: 10.1200/EDBK_349799. Am Soc Clin Oncol Educ Book. 2022. PMID: 35580293
-
Immune checkpoint inhibitors in gastrointestinal malignancies: an Umbrella review.Cancer Cell Int. 2024 Jan 5;24(1):10. doi: 10.1186/s12935-023-03183-3. Cancer Cell Int. 2024. PMID: 38183112 Free PMC article. Review.
-
Immune checkpoint inhibitors therapy as the game-changing approach for pediatric lymphoma: A brief landscape.Crit Rev Oncol Hematol. 2024 Jan;193:104225. doi: 10.1016/j.critrevonc.2023.104225. Epub 2023 Dec 3. Crit Rev Oncol Hematol. 2024. PMID: 38049077 Review.
-
Efficacy and Safety of Immune Checkpoint Inhibitors in Hematologic Malignancies.Clin Lymphoma Myeloma Leuk. 2024 Jan;24(1):23-31. doi: 10.1016/j.clml.2023.09.002. Epub 2023 Sep 19. Clin Lymphoma Myeloma Leuk. 2024. PMID: 37863681 Review.
-
The Challenge of Developing New Therapies for Childhood Cancers.Oncologist. 1997;2(1):I-II. Oncologist. 1997. PMID: 10388032
Cited by
-
Alopecia in Children with Cancer: A Review from Pathophysiology to Management.Am J Clin Dermatol. 2025 Jun 30. doi: 10.1007/s40257-025-00960-w. Online ahead of print. Am J Clin Dermatol. 2025. PMID: 40587083 Review.
-
Overcoming the blood-brain barrier (BBB) in pediatric CNS tumors: immunotherapy and nanomedicine-driven strategies.Med Oncol. 2025 Aug 19;42(10):431. doi: 10.1007/s12032-025-02984-y. Med Oncol. 2025. PMID: 40828479 Review.
References
-
- Xia L., Zhou Q., Gao Y., Hu W., Lou G., Sun H., Zhu J., Shu J., Zhou X., Sun R., et al. A multicenter phase 2 trial of camrelizumab plus famitinib for women with recurrent or metastatic cervical squamous cell carcinoma. Nat. Commun. 2022;13:7581. doi: 10.1038/s41467-022-35133-4. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

