Design and Fabrication of Sustained Bacterial Release Scaffolds to Support the Microbiome
- PMID: 39204410
- PMCID: PMC11358975
- DOI: 10.3390/pharmaceutics16081066
Design and Fabrication of Sustained Bacterial Release Scaffolds to Support the Microbiome
Abstract
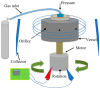
Fibres in the micro- and nanometre scale are suited to a broad range of applications, including drug delivery and tissue engineering. Electrospinning is the manufacturing method of choice, but it has some limitations. Novel pressure-driven fibre-forming techniques, like pressurised gyration (PG), overcome these limitations; however, the compatibility of PG with biological materials has not yet been evaluated in detail. For the first time, this limitation of PG was investigated by optimising PG for microbial cell processing and incorporating bacterial cultures into fibrous polymeric scaffolds for sustained release. Multiple polymer-solvent systems were trialled, including polyvinylpyrrolidone (PVP)/phosphate-buffered saline (PBS) 25% w/v, polyethylene oxide (PEO)/PBS 20% w/v, and PVP/ethanol 20% w/v. Rheological studies revealed the surface tension of the PVP/PBS, PEO/PBS, and PVP/ethanol polymer-solvent systems to be 73.2, 73.9, and 22.6 mN/m, respectively. Scanning electron microscopy showed the median fibre diameters to be between 9.8 μm and 26.1 μm, with PVP producing larger fibres. Overnight Bacillus subtilis cultures were then incorporated into the chosen polymeric solutions and processed into fibres using PG. The produced cell-loaded fibres were incubated in LB broth to assess the cell viability of the encapsulated cells. Colony counts post-incubation showed the PVP/PBS 25% fibres resulted in 60% bacterial growth, and PEO/PBS 20% fibres led to 47% bacterial growth, whereas PVP/ethanol 20% fibres did not lead to any bacterial growth. Based on the results gathered during this study, it can be concluded that PG offers a promising way of encapsulating cells and other sensitive biological products while having many notable advantages compared to electrospinning. This research demonstrates proof of concept research-based evidence and showcases the potential of pressurised gyration as a key disruptive innovation in probiotic delivery system design and manufacturing.
Keywords: bioactive fibres; drug delivery; microfibres; probiotics; sustained release.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Electrospun PVP-indomethacin constituents for transdermal dressings and drug delivery devices.Int J Pharm. 2014 Oct 1;473(1-2):95-104. doi: 10.1016/j.ijpharm.2014.06.059. Epub 2014 Jul 2. Int J Pharm. 2014. PMID: 24997411
-
Fabrication of multilayered electrospun poly(lactic-co-glycolic acid)/polyvinyl pyrrolidone + poly(ethylene oxide) scaffolds and biocompatibility evaluation.J Biomed Mater Res A. 2021 Aug;109(8):1468-1478. doi: 10.1002/jbm.a.37137. Epub 2020 Dec 9. J Biomed Mater Res A. 2021. PMID: 33289293
-
Electrospinning and evaluation of PHBV-based tissue engineering scaffolds with different fibre diameters, surface topography and compositions.J Biomater Sci Polym Ed. 2012;23(6):779-806. doi: 10.1163/092050611X560708. J Biomater Sci Polym Ed. 2012. PMID: 21418747
-
Bacterial cellulose micro-nano fibres for wound healing applications.Biotechnol Adv. 2020 Jul-Aug;41:107549. doi: 10.1016/j.biotechadv.2020.107549. Epub 2020 Apr 14. Biotechnol Adv. 2020. PMID: 32302653 Review.
-
Pharmaceutical assessment of polyvinylpyrrolidone (PVP): As excipient from conventional to controlled delivery systems with a spotlight on COVID-19 inhibition.J Drug Deliv Sci Technol. 2020 Dec;60:102046. doi: 10.1016/j.jddst.2020.102046. Epub 2020 Sep 2. J Drug Deliv Sci Technol. 2020. PMID: 32905026 Free PMC article. Review.
References
-
- Leena M.M., Yoha K.S., Moses J.A., Anandharamakrishnan C. Innovative Food Processing Technologies. Elsevier; Amsterdam, The Netherlands: 2021. Nanofibers in Food Applications; pp. 634–650.
Grants and funding
LinkOut - more resources
Full Text Sources

