New Insights into the Connections between Flooding/Hypoxia Response and Plant Defenses against Pathogens
- PMID: 39204612
- PMCID: PMC11358971
- DOI: 10.3390/plants13162176
New Insights into the Connections between Flooding/Hypoxia Response and Plant Defenses against Pathogens
Abstract
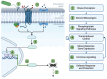
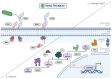
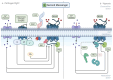
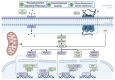
The impact of global climate change has highlighted the need for a better understanding of how plants respond to multiple simultaneous or sequential stresses, not only to gain fundamental knowledge of how plants integrate signals and mount a coordinated response to stresses but also for applications to improve crop resilience to environmental stresses. In recent years, there has been a stronger emphasis on understanding how plants integrate stresses and the molecular mechanisms underlying the crosstalk between the signaling pathways and transcriptional programs that underpin plant responses to multiple stresses. The combination of flooding (or resulting hypoxic stress) with pathogen infection is particularly relevant due to the frequent co-occurrence of both stresses in nature. This review focuses on (i) experimental approaches and challenges associated with the study of combined and sequential flooding/hypoxia and pathogen infection, (ii) how flooding (or resulting hypoxic stress) influences plant immunity and defense responses to pathogens, and (iii) how flooding contributes to shaping the soil microbiome and is linked to plants' ability to fight pathogen infection.
Keywords: combined stress; flooding; hypoxia; plant defenses against pathogens; plant immunity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
'Against all floods': plant adaptation to flooding stress and combined abiotic stresses.Plant J. 2024 Mar;117(6):1836-1855. doi: 10.1111/tpj.16614. Epub 2024 Jan 13. Plant J. 2024. PMID: 38217848 Review.
-
Flooding stress and responses to hypoxia in plants.Funct Plant Biol. 2024 Mar;51:FP24061. doi: 10.1071/FP24061. Funct Plant Biol. 2024. PMID: 38538565
-
Plant Immune System: Crosstalk Between Responses to Biotic and Abiotic Stresses the Missing Link in Understanding Plant Defence.Curr Issues Mol Biol. 2017;23:1-16. doi: 10.21775/cimb.023.001. Epub 2017 Feb 3. Curr Issues Mol Biol. 2017. PMID: 28154243 Review.
-
Elucidating the Response of Crop Plants towards Individual, Combined and Sequentially Occurring Abiotic Stresses.Int J Mol Sci. 2021 Jun 6;22(11):6119. doi: 10.3390/ijms22116119. Int J Mol Sci. 2021. PMID: 34204152 Free PMC article. Review.
-
Microbiome structure variation and soybean's defense responses during flooding stress and elevated CO2.Front Plant Sci. 2024 Feb 8;14:1295674. doi: 10.3389/fpls.2023.1295674. eCollection 2023. Front Plant Sci. 2024. PMID: 38389716 Free PMC article.
Cited by
-
Double-truncated version of OsGADs leads to higher GABA accumulation and stronger stress tolerance in Oryza sativa L. var. japonica.Plant Cell Rep. 2025 Apr 8;44(5):95. doi: 10.1007/s00299-025-03477-y. Plant Cell Rep. 2025. PMID: 40198389 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

