Patients with Waldenström macroglobulinemia have impaired platelet and coagulation function
- PMID: 39207869
- PMCID: PMC11539362
- DOI: 10.1182/bloodadvances.2024014190
Patients with Waldenström macroglobulinemia have impaired platelet and coagulation function
Abstract
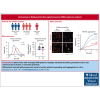
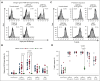
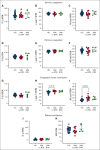
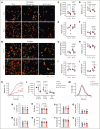
Clinical features in patients with the B-cell lymphoma, Waldenström macroglobulinemia (WM), include cytopenias, immunoglobulin M (IgM)-mediated hyperviscosity, fatigue, bleeding, and bruising. Therapeutics such as Bruton's tyrosine kinase inhibitors (BTKis) exacerbate bleeding risk. Abnormal hemostasis arising from platelet dysfunction, altered coagulation or vascular impairment have not yet been investigated in patients with WM. Therefore, this study aimed to evaluate hemostatic dysfunction in samples from these patients. Whole blood (WB) samples were collected from 14 patients with WM not receiving therapy, 5 patients receiving BTKis and 15 healthy donors (HDs). Platelet receptor levels and reticulation were measured by flow cytometry, plasma thrombin generation with or without platelets by fluorescence resonance energy transfer assay, WB clotting potential by rotational thromboelastometry, and plasma soluble glycoprotein VI (sGPVI) and serum thrombopoietin (TPO) by enzyme-linked immunosorbent assay. Donor platelet spreading, aggregation, and ability to accelerate thrombin generation in the presence of WM-derived IgM were assessed. WM platelet receptor levels, responses to physiological agonists, and plasma sGPVI were within normal ranges. WM platelets had reduced reticulation (P = .0012) whereas serum TPO levels were increased (P = .0040). WM plasma displayed slower thrombin generation (P = .0080) and WM platelets contributed less to endogenous thrombin potential (ETP; P = .0312). HD plasma or platelets incubated with IgM (50-60 mg/mL) displayed reduced spreading (P = .0002), aggregation (P < .0001), and ETP (P = .0081). Thus, alterations to thrombin potential and WB coagulation were detected in WM samples. WM IgM significantly impaired hemostasis in vitro. Platelet and coagulation properties are disturbed in patients with well-managed WM.
© 2024 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures


References
-
- Sekhar J, Sanfilippo K, Zhang Q, Trinkaus K, Vij R, Morgensztern D. Waldenström macroglobulinemia: a surveillance, epidemiology, and end results database review from 1988 to 2005. Leuk Lymphoma. 2012;53(8):1625–1626. - PubMed
-
- Pophali PA, Bartley A, Kapoor P, et al. Prevalence and survival of smouldering Waldenström macroglobulinaemia in the United States. Br J Haematol. 2019;184(6):1014–1017. - PubMed
-
- Talaulikar D, Joshua D, Ho PJ, et al. Treatment of patients with Waldenström macroglobulinaemia: clinical practice update from the Myeloma Foundation of Australia Medical and Scientific Advisory Group. Intern Med J. 2023;53(4):599–609. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

