Activation mechanisms of dimeric mechanosensitive OSCA/TMEM63 channels
- PMID: 39209849
- PMCID: PMC11362595
- DOI: 10.1038/s41467-024-51800-0
Activation mechanisms of dimeric mechanosensitive OSCA/TMEM63 channels
Abstract
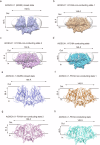
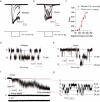
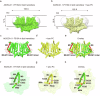
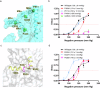
OSCA/TMEM63 channels, which have transporter-like architectures, are bona fide mechanosensitive (MS) ion channels that sense high-threshold mechanical forces in eukaryotic cells. The activation mechanism of these transporter-like channels is not fully understood. Here we report cryo-EM structures of a dimeric OSCA/TMEM63 pore mutant OSCA1.1-F516A with a sequentially extracellular dilated pore in a detergent environment. These structures suggest that the extracellular pore sequential dilation resembles a flower blooming and couples to a sequential contraction of each monomer subunit towards the dimer interface and subsequent extrusion of the dimer interface lipids. Interestingly, while OSCA1.1-F516A remains non-conducting in the native lipid environment, it can be directly activated by lyso-phosphatidylcholine (Lyso-PC) with reduced single-channel conductance. Structural analysis of OSCA1.1-F516A in lyso-PC-free and lyso-PC-containing lipid nanodiscs indicates that lyso-PC induces intracellular pore dilation by attracting the M6b to upward movement away from the intracellular side thus extending the intracellular pore. Further functional studies indicate that full activation of MS OSCA/TMEM63 dimeric channels by high-threshold mechanical force also involves the opening of both intercellular and extracellular pores. Our results provide the fundamental activation paradigm of the unique transporter-like MS OSCA/TMEM63 channels, which is likely applicable to functional branches of the TMEM63/TMEM16/TMC superfamilies.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

