A bioactive composite scaffold enhances osteochondral repair by using thermosensitive chitosan hydrogel and endothelial lineage cell-derived chondrogenic cell
- PMID: 39211289
- PMCID: PMC11357856
- DOI: 10.1016/j.mtbio.2024.101174
A bioactive composite scaffold enhances osteochondral repair by using thermosensitive chitosan hydrogel and endothelial lineage cell-derived chondrogenic cell
Abstract
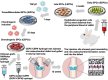
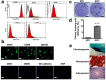
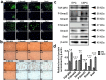
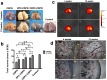
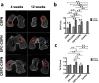
Articular cartilage regeneration is a major challenge in orthopedic medicine. Endothelial progenitor cells (EPCs) are a promising cell source for regenerative medicine applications. However, their roles and functions in cartilage regeneration are not well understood. Additionally, thermosensitive chitosan hydrogels have been widely used in tissue engineering, but further development of these hydrogels incorporating vascular lineage cells for cartilage repair is insufficient. Thus, this study aimed to characterize the ability of EPCs to undergo endothelial-mesenchymal stem cell transdifferentiation and chondrogenic differentiation and investigate the ability of chondrogenic EPC-seeded thermosensitive chitosan-graft-poly (N-isopropylacrylamide) (CEPC-CSPN) scaffolds to improve healing in a rabbit osteochondral defect (OCD) model. EPCs were isolated and endothelial-to-mesenchymal transition (EndMT) was induced by transforming growth factor-β1 (TGF-β1); these EPCs are subsequently termed transdifferentiated EPCs (tEPCs). The stem cell-like properties and chondrogenic potential of tEPCs were evaluated by a series of in vitro assays. Furthermore, the effect of CEPC-CSPN scaffolds on OCD repair was evaluated. Our in vitro results confirmed that treatment of EPC with TGF-β1 induced EndMT and the acquisition of stem cell-like properties, producing tEPCs. Upon inducing chondrogenic differentiation of tEPCs (CEPCs), the cells exhibited significantly enhanced chondrogenesis and chondrocyte surface markers after 25 days. The TGF-β1-induced differentiation of EPCs is mediated by both the TGF-β/Smad and extracellular signal-regulated kinase (Erk) pathways. The CEPC-CSPN scaffold reconstructed well-integrated translucent cartilage and repaired subchondral bone in vivo, exhibiting regenerative capacity. Collectively, our results suggest that the CEPC-CSPN scaffold induces OCD repair, representing a promising approach to articular cartilage regeneration.
Keywords: Chondrogenesis; Endothelial progenitor cells; Endothelial-to-mesenchymal transition; Osteochondral regeneration; Thermosensitive injectable hydrogels.
© 2024 Published by Elsevier Ltd.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




Similar articles
-
Acceleration of chondrogenic differentiation of human mesenchymal stem cells by sustained growth factor release in 3D graphene oxide incorporated hydrogels.Acta Biomater. 2020 Mar 15;105:44-55. doi: 10.1016/j.actbio.2020.01.048. Epub 2020 Feb 5. Acta Biomater. 2020. PMID: 32035282
-
Repair of osteochondral defects using injectable chitosan-based hydrogel encapsulated synovial fluid-derived mesenchymal stem cells in a rabbit model.Mater Sci Eng C Mater Biol Appl. 2019 Jun;99:541-551. doi: 10.1016/j.msec.2019.01.115. Epub 2019 Jan 26. Mater Sci Eng C Mater Biol Appl. 2019. PMID: 30889728
-
Nanoscale Thermosensitive Hydrogel Scaffolds Promote the Chondrogenic Differentiation of Dental Pulp Stem and Progenitor Cells: A Minimally Invasive Approach for Cartilage Regeneration.Int J Nanomedicine. 2020 Oct 12;15:7775-7789. doi: 10.2147/IJN.S274418. eCollection 2020. Int J Nanomedicine. 2020. PMID: 33116500 Free PMC article.
-
Chondrogenic differentiation of mesenchymal stem cells and its clinical applications.Yonsei Med J. 2004 Jun 30;45 Suppl:41-7. doi: 10.3349/ymj.2004.45.Suppl.41. Yonsei Med J. 2004. PMID: 15250049 Review.
-
Osteochondral Tissue Engineering Dilemma: Scaffolding Trends in Regenerative Medicine.Stem Cell Rev Rep. 2023 Aug;19(6):1615-1634. doi: 10.1007/s12015-023-10545-x. Epub 2023 Apr 19. Stem Cell Rev Rep. 2023. PMID: 37074547 Review.
Cited by
-
Injectable Biopolymer-Based Hydrogels: A Next-Generation Platform for Minimally Invasive Therapeutics.Gels. 2025 May 23;11(6):383. doi: 10.3390/gels11060383. Gels. 2025. PMID: 40558682 Free PMC article. Review.
-
Current Utilization of Gel-Based Scaffolds and Templates in Foot and Ankle Surgery-A Review.Gels. 2025 Apr 24;11(5):316. doi: 10.3390/gels11050316. Gels. 2025. PMID: 40422336 Free PMC article. Review.
References
-
- Wang H.C., Lin T.H., Chang N.J., Hsu H.C., Yeh M.L. Continuous passive motion promotes and maintains chondrogenesis in autologous endothelial progenitor cell-loaded porous PLGA scaffolds during osteochondral defect repair in a rabbit model. Int. J. Mol. Sci. 2019;20 doi: 10.3390/ijms20020259. - DOI - PMC - PubMed
-
- Hur J., Yoon C.H., Kim H.S., Choi J.H., Kang H.J., Hwang K.K., Oh B.H., Lee M.M., Park Y.B. Characterization of two types of endothelial progenitor cells and their different contributions to neovasculogenesis. Arterioscler. Thromb. Vasc. Biol. 2004;24:288–293. doi: 10.1161/01.atv.0000114236.77009.06. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

