Routes and molecular mechanisms of central nervous system involvement in acute myeloid leukemia (Review)
- PMID: 39219268
- PMCID: PMC11378150
- DOI: 10.3892/or.2024.8805
Routes and molecular mechanisms of central nervous system involvement in acute myeloid leukemia (Review)
Abstract
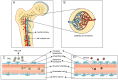
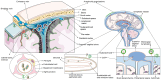
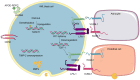
Acute myeloid leukemia (AML) is a predominant form of leukemia. Central nervous system (CNS) involvement complicates its diagnosis due to limited diagnostic tools, as well as its treatment due to inadequate therapeutic methodologies and poor prognosis. Furthermore, its incidence rate is unclear. The mechanisms of AML cell mobilization from the bone marrow (BM) to the CNS are not fully elucidated, and the molecular factors contributing to CNS infiltration are insufficiently recognized. The present review aimed to enhance the understanding of CNS involvement of AML and its impact on CNS. The latest research on the pathways and mechanisms facilitating AML cells to escape the BM and infiltrate the CNS was reviewed. Additionally, novel therapeutic strategies targeting specific molecules and genes for treating CNS involvement in AML were examined.
Keywords: AML; CNS; brain barrier; cell adhesion molecules; therapeutic strategies.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
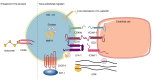
Similar articles
-
Central Nervous System Involvement at the Time of Allogeneic Hematopoietic Stem Cell Transplantation Is Associated with a Poor Outcome in Patients with Acute Myeloid Leukemia.Pathol Oncol Res. 2017 Apr;23(2):433-437. doi: 10.1007/s12253-016-0162-6. Epub 2016 Dec 21. Pathol Oncol Res. 2017. PMID: 28004352
-
Tailored central nervous system-directed treatment strategy for isolated CNS recurrence of adult acute myeloid leukemia.Hematology. 2014 Jun;19(4):208-12. doi: 10.1179/1607845413Y.0000000117. Epub 2013 Nov 25. Hematology. 2014. PMID: 24074528
-
The association of leukocyte immunoglobulin-like receptor subfamily B-4 expression in acute myeloid leukemia and central nervous system involvement.Leuk Res. 2021 Jan;100:106480. doi: 10.1016/j.leukres.2020.106480. Epub 2020 Nov 22. Leuk Res. 2021. PMID: 33285315
-
Systematic review and meta-analysis evaluating clinical outcomes in adult acute myeloid leukemia patients with central nervous system involvement.Leuk Res. 2024 Feb;137:107452. doi: 10.1016/j.leukres.2024.107452. Epub 2024 Feb 6. Leuk Res. 2024. PMID: 38335816
-
Roles of the bone marrow niche in hematopoiesis, leukemogenesis, and chemotherapy resistance in acute myeloid leukemia.Hematology. 2018 Dec;23(10):729-739. doi: 10.1080/10245332.2018.1486064. Epub 2018 Jun 14. Hematology. 2018. PMID: 29902132 Review.
References
-
- Munker R, Labopin M, Esteve J, Schmid C, Mohty M, Nagler A. Mixed phenotype acute leukemia: Outcomes with allogeneic stem cell transplantation. A retrospective study from the Acute Leukemia Working Party of the EBMT. Haematologica. 2017;102:2134–2140. doi: 10.3324/haematol.2017.174441. - DOI - PMC - PubMed
-
- Dima D, Oprita L, Rosu AM, Trifa A, Selicean C, Moisoiu V, Frinc I, Zdrenghea M, Tomuleasa C. Adult acute megakaryoblastic leukemia: Rare association with cytopenias of undetermined significance and p210 and p190 BCR-ABL transcripts. Onco Targets Ther. 2017;10:5047–5051. doi: 10.2147/OTT.S146973. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

