3D-printed aerogels as theranostic implants monitored by fluorescence bioimaging
- PMID: 39220405
- PMCID: PMC11364008
- DOI: 10.1016/j.bioactmat.2024.07.033
3D-printed aerogels as theranostic implants monitored by fluorescence bioimaging
Abstract
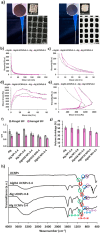
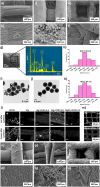
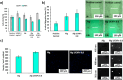
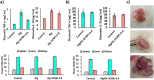
Aerogel scaffolds are nanostructured materials with beneficial properties for tissue engineering applications. The tracing of the state of the aerogels after their implantation is challenging due to their variable biodegradation rate and the lack of suitable strategies capable of in vivo monitoring the scaffolds. Upconversion nanoparticles (UCNPs) have emerged as advanced tools for in vitro bioimaging because of their fluorescence properties. In this work, highly fluorescent UCNPs were loaded into aerogels to obtain theranostic implants for tissue engineering and bioimaging applications. 3D-printed alginate-hydroxyapatite aerogels labeled with UCNPs were manufactured by 3D-printing and supercritical CO2 drying to generate personalize-to-patient aerogels. The physicochemical performance of the resulting structures was evaluated by printing fidelity measurements, nitrogen adsorption-desorption analysis, and different microscopies (confocal, transmission and scanning electron microscopies). Stability of the aerogels in terms of physicochemical properties was also tested after 3 years of storage. Biocompatibility was evaluated in vitro by different cell and hemocompatibility assays, in ovo and in vivo by safety and bioimaging studies using different murine models. Cytokines profile, tissue index and histological evaluations of the main organs unveiled an in vivo downregulation of the inflammation after implantation of the scaffolds. UCNPs-decorated aerogels were first-time manufactured and long-term traceable by fluorescence-based bioimaging until 3 weeks post-implantation, thereby endorsing their suitability as tissue engineering and theranostic nanodevices (i.e. bifunctional implants).
Keywords: Aerogels; In vivo fluorescence; Theranostic implants; Upconversion nanoparticles.
© 2024 The Authors.
Conflict of interest statement
All authors disclose no actual or potential conflict of interest related with any financial and personal relationships with other people or organizations that could inappropriately influence, or be perceived to influence, this work.
Figures




References
-
- Yamaoka T. In: Encyclopedia of Polymeric Nanomaterials. Kobayashi S., Müllen K., editors. Springer Berlin Heidelberg; Berlin, Heidelberg: 2014. Bioimaging materials; pp. 1–6. - DOI
LinkOut - more resources
Full Text Sources

