Evaluation of ABD-Linked RM26 Conjugates for GRPR-Targeted Drug Delivery
- PMID: 39220525
- PMCID: PMC11359615
- DOI: 10.1021/acsomega.4c00489
Evaluation of ABD-Linked RM26 Conjugates for GRPR-Targeted Drug Delivery
Abstract
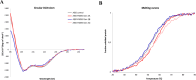
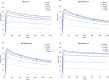
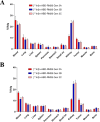
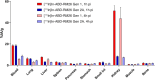
Targeting the gastrin-releasing peptide receptor (GRPR) with the bombesin analogue RM26, a 9 aa peptide, has been a promising strategy for cancer theranostics, with recent success in radionuclide imaging of prostate cancer. However, therapeutic application of the short peptide RM26 would require a longer half-life to prevent fast clearance from the circulation. Conjugation to an albumin-binding domain (ABD) is a viable strategy to extend the in vivo half-life of peptides and proteins. We previously reported an ABD-fused RM26 peptide targeting GRPR (ABD-RM26 Gen 1) that showed prolonged and stable tumor uptake over 144 h; however, the observed high kidney uptake indicated that the conjugate's binding to albumin was reduced and that this could be an obstacle for its use as a delivery system for targeted therapy, especially for radiotherapy. Here, we have designed, produced, and preclinically evaluated a series of novel ABD-RM26 conjugates with the aim of improving the conjugate's binding to albumin and decreasing the kidney uptake. We developed three second-generation constructs with varying formats, differing in the relative positions of the targeting moieties and the radionuclide chelator. The produced conjugates were radiolabeled with indium-111 and evaluated in vitro and in vivo. All constructs displayed improved biophysical characteristics, biodistribution, and lower kidney uptake compared to previously reported first-generation molecules. The ABD-RM26 Gen 2A conjugate showed the best biodistribution profile with a nearly 6-fold reduction in kidney uptake. However, the ABD-RM26 Gen 2A conjugate's binding to GRPR was compromised. This conjugate's assembly of albumin- and GRPR-binding moieties might be used for further development of drug conjugates for targeted therapy/radiotherapy of GRPR-expressing cancers.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Preclinical Evaluation of the GRPR-Targeting Antagonist RM26 Conjugated to the Albumin-Binding Domain for GRPR-Targeting Therapy of Cancer.Pharmaceutics. 2020 Oct 16;12(10):977. doi: 10.3390/pharmaceutics12100977. Pharmaceutics. 2020. PMID: 33081166 Free PMC article.
-
The effect of macrocyclic chelators on the targeting properties of the 68Ga-labeled gastrin releasing peptide receptor antagonist PEG2-RM26.Nucl Med Biol. 2015 May;42(5):446-454. doi: 10.1016/j.nucmedbio.2014.12.009. Epub 2014 Dec 20. Nucl Med Biol. 2015. PMID: 25684649
-
Selection of optimal chelator improves the contrast of GRPR imaging using bombesin analogue RM26.Int J Oncol. 2016 May;48(5):2124-34. doi: 10.3892/ijo.2016.3429. Epub 2016 Mar 9. Int J Oncol. 2016. PMID: 26983776
-
Preclinical Evaluation of the Copper-64 Labeled GRPR-Antagonist RM26 in Comparison with the Cobalt-55 Labeled Counterpart for PET-Imaging of Prostate Cancer.Molecules. 2020 Dec 18;25(24):5993. doi: 10.3390/molecules25245993. Molecules. 2020. PMID: 33352838 Free PMC article.
-
GRPr Theranostics: Current Status of Imaging and Therapy using GRPr Targeting Radiopharmaceuticals.Nuklearmedizin. 2022 Jun;61(3):247-261. doi: 10.1055/a-1759-4189. Epub 2022 Jun 3. Nuklearmedizin. 2022. PMID: 35668669 Review. English.
Cited by
-
Targeting of Epithelial Cell Adhesion Molecule-Expressing Malignant Tumors Using an Albumin-Binding Domain-Fused Designed Ankyrin Repeat Protein: Effect of the Molecular Architecture.Int J Mol Sci. 2025 May 29;26(11):5236. doi: 10.3390/ijms26115236. Int J Mol Sci. 2025. PMID: 40508045 Free PMC article.
-
A preclinical study on the influence of linkers in [68Ga]Ga-NOTA-X-RM26 radiotracers for PET imaging of GRPR expression.EJNMMI Res. 2025 Aug 7;15(1):104. doi: 10.1186/s13550-025-01301-y. EJNMMI Res. 2025. PMID: 40775579 Free PMC article.
References
-
- Markwalder R.; Reubi J. C. Gastrin-releasing Peptide Receptors in the Human Prostate: Relation to Neoplastic Transformation. Cancer Res. 1999, 59 (5), 1152–1159. - PubMed
LinkOut - more resources
Full Text Sources
