Design considerations for digital light processing bioprinters
- PMID: 39221036
- PMCID: PMC11284760
- DOI: 10.1063/5.0187558
Design considerations for digital light processing bioprinters
Abstract
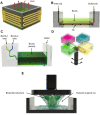
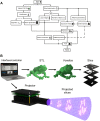
With the rapid development and popularization of additive manufacturing, different technologies, including, but not limited to, extrusion-, droplet-, and vat-photopolymerization-based fabrication techniques, have emerged that have allowed tremendous progress in three-dimensional (3D) printing in the past decades. Bioprinting, typically using living cells and/or biomaterials conformed by different printing modalities, has produced functional tissues. As a subclass of vat-photopolymerization bioprinting, digital light processing (DLP) uses digitally controlled photomasks to selectively solidify liquid photocurable bioinks to construct complex physical objects in a layer-by-layer manner. DLP bioprinting presents unique advantages, including short printing times, relatively low manufacturing costs, and decently high resolutions, allowing users to achieve significant progress in the bioprinting of tissue-like complex structures. Nevertheless, the need to accommodate different materials while bioprinting and improve the printing performance has driven the rapid progress in DLP bioprinters, which requires multiple pieces of knowledge ranging from optics, electronics, software, and materials beyond the biological aspects. This raises the need for a comprehensive review to recapitulate the most important considerations in the design and assembly of DLP bioprinters. This review begins with analyzing unique considerations and specific examples in the hardware, including the resin vat, optical system, and electronics. In the software, the workflow is analyzed, including the parameters to be considered for the control of the bioprinter and the voxelizing/slicing algorithm. In addition, we briefly discuss the material requirements for DLP bioprinting. Then, we provide a section with best practices and maintenance of a do-it-yourself DLP bioprinter. Finally, we highlight the future outlooks of the DLP technology and their critical role in directing the future of bioprinting. The state-of-the-art progress in DLP bioprinter in this review will provide a set of knowledge for innovative DLP bioprinter designs.
© 2024 Author(s).
Conflict of interest statement
YSZ consulted for Allevi by 3D systems, and sits on the scientific advisory board and holds options of Xellar, neither of which, however, participated in or bias the work. The interests are managed by the Brigham and Women's Hospital. The other authors declare no COI.
Figures






References
-
- Wohlers T. and Gornet T., History of Additive Manufacturing ( Wohlers Associates Inc., 2014).
-
- Ngo T. D., Kashani A., Imbalzano G., Nguyen K. T. Q., and Hui D., “ Additive manufacturing (3D printing): A review of materials, methods, applications and challenges,” Compos. B Eng. 143, 172–196 (2018). 10.1016/j.compositesb.2018.02.012 - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
