Cytokine release syndrome following COVID-19 infection during treatment with nivolumab for cancer of esophagogastric junction carcinoma: a case report and review
- PMID: 39223460
- PMCID: PMC11367929
- DOI: 10.1186/s12245-024-00691-5
Cytokine release syndrome following COVID-19 infection during treatment with nivolumab for cancer of esophagogastric junction carcinoma: a case report and review
Abstract
Background: Cytokine release syndrome (CRS) is an acute systemic inflammatory syndrome characterized by fever and multiple organ failure, which is triggered by immunotherapy or certain infections. Immune checkpoint inhibitors rarely cause immune-related adverse event- cytokine release syndrome (irAE-CRS). This article presents a case report of irAE-CRS triggered by coronavirus disease 2019 (COVID-19).
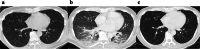
Case presentation: A 60-year-old man with type 2 diabetes received nivolumab treatment for esophagogastric junction carcinoma and experienced two immune-related adverse events: hypothyroidism and skin disorder. Eleven days before his visit to our hospital, he had a fever and was diagnosed with COVID-19. Five days before his visit, he developed a fever again, along with general malaise, water soluble diarrhea, and myalgia of the extremities. On admission, the patient was in a state of multiple organ failure, and although the source of infection was unknown, a tentative diagnosis of septic shock was made. The patient's condition was unstable despite systemic management with antimicrobial agents, high-dose vasopressors, and intravenous fluids. We suspected CRS due to irAE (irAE-CRS) based on his history of nivolumab use. Steroid pulse therapy (methylprednisolone 1 g/day) was started, and the patient temporarily recovered. However, his respiratory condition worsened; consequently, he was placed on a ventilator and tocilizumab was added to the treatment. His muscle strength recovered to the point where he could live at home, and was subsequently discharged.
Conclusion: In patients previously treated with immune checkpoint inhibitors, irAE-CRS should be considered as a differential diagnosis when multiple organ damage is observed in addition to inflammatory findings. It is recommended to start treatment with steroids; if the disease is refractory, other immunosuppressive therapies such as tocilizumab should be introduced as early as possible.
Keywords: Cytokine release syndrome; Immune checkpoint inhibitor; Immune-related adverse event.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Honjo O, Kubo T, Sugaya F, Nishizaka T, Kato K, Hirohashi Y, et al. Severe cytokine release syndrome resulting in purpura fulminans despite successful response to nivolumab therapy in a patient with pleomorphic carcinoma of the lung: a case report. J Immunother Cancer. 2019;7:97. 10.1186/s40425-019-0582-4 - DOI - PMC - PubMed
-
- Carvajal RD, Sato T, Butler MO, Sacco JJ, Shoushtari AN, Hassel JC, et al. Characterization of cytokine release syndrome (CRS) following treatment with tebentafusp in patients (pts) with previously treated (2L+) metastatic uveal melanoma(mUM). J Clin Oncol. 2021. 10.1200/JCO.2021.39.15_suppl.9531. 10.1200/JCO.2021.39.15_suppl.9531 - DOI - PubMed
LinkOut - more resources
Full Text Sources

