Giant viruses inhibit superinfection by downregulating phagocytosis in Acanthamoeba
- PMID: 39225468
- PMCID: PMC11494976
- DOI: 10.1128/jvi.01045-24
Giant viruses inhibit superinfection by downregulating phagocytosis in Acanthamoeba
Abstract
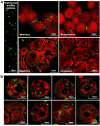
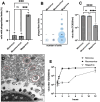
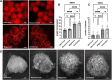
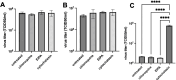
In the context of the virosphere, viral particles can compete for host cells. In this scenario, some viruses block the entry of exogenous virions upon infecting a cell, a phenomenon known as superinfection inhibition. The molecular mechanisms associated with superinfection inhibition vary depending on the viral species and the host, but generally, blocking superinfection ensures the genetic supremacy of the virus's progeny that first infects the cell. Giant amoeba-infecting viruses have attracted the scientific community's attention due to the complexity of their particles and genomes. However, there are no studies on the occurrence of superinfection and its inhibition induced by giant viruses. This study shows that mimivirus, moumouvirus, and megavirus, exhibit different strategies related to the infection of Acanthamoeba. For the first time, we have reported that mimivirus and moumouvirus induce superinfection inhibition in amoebas. Interestingly, megaviruses do not exhibit this ability, allowing continuous entry of exogenous virions into infected amoebas. Our investigation into the mechanisms behind superinfection blockage reveals that mimivirus and moumouvirus inhibit amoebic phagocytosis, leading to significant changes in the morphology and activity of the host cells. In contrast, megavirus-infected amoebas continue incorporating newly formed virions, negatively affecting the available viral progeny. This effect, however, is reversible with chemical inhibition of phagocytosis. This work contributes to the understanding of superinfection and its inhibition in mimivirus, moumouvirus, and megavirus, demonstrating that despite their evolutionary relatedness, these viruses exhibit profound differences in their interactions with their hosts.IMPORTANCESome viruses block the entry of new virions upon infecting a cell, a phenomenon known as superinfection inhibition. Superinfection inhibition in giant viruses has yet to be studied. This study reveals that even closely related viruses, such as mimivirus, moumouvirus, and megavirus, have different infection strategies for Acanthamoeba. For the first time, we have reported that mimivirus and moumouvirus induce superinfection inhibition in amoebas. In contrast, megaviruses do not exhibit this ability, allowing continuous entry of exogenous virions into infected amoebas. Our investigation shows that mimivirus and moumouvirus inhibit amoebic phagocytosis, causing significant changes in host cell morphology and activity. Megavirus-infected amoebas, however, continue incorporating newly formed viruses, affecting viral progeny. This research enhances our understanding of superinfection inhibition in these viruses, highlighting their differences in host interactions.
Keywords: Acanthamoeba; giant virus; phagocytosis; superinfection; virus-host relationship.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Schwartz EJ, Biggs KRH, Bailes C, Ferolito KA, Vaidya NK. 2016. HIV dynamics with immune responses: perspectives from mathematical modeling. Curr Clin Microbiol Rep 3:216–224. doi:10.1007/s40588-016-0049-z - DOI
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

