This is a preprint.
Microenvironment T-Type calcium channels regulate neuronal and glial processes to promote glioblastoma growth
- PMID: 39229003
- PMCID: PMC11370607
- DOI: 10.1101/2024.08.22.609229
Microenvironment T-Type calcium channels regulate neuronal and glial processes to promote glioblastoma growth
Abstract
Background: Glioblastoma (GBM) is the most common primary malignant brain tumor. The aim of this study was to elucidate the role of microenvironment and intrinsic T-type calcium channels (Cav3) in regulating tumor growth and progression.
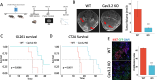
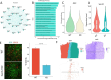
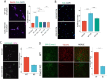
Methods: We grafted syngeneic GBM cells into Cav3.2 knockout mice to assess the role of microenvironment T-Type calcium channels on GBM tumor growth. We performed single-cell RNA-seq (scRNA-seq) of tumors from WT and Cav3.2 KO mice to elucidate the regulation of tumors by the microenvironment. We used neurons from WT and Cav3.2 KO mice in co-culture with GBM stem cells (GSC) to assess the effects of Cav3.2 on neuron/GSC synaptic connections and tumor cell growth.
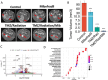
Results: Cav3.2 KO in the microenvironment led to significant reduction of GBM growth and prolongation of animal survival. scRNA-seq showed that microenvironment Cav3.2 regulates neuronal and glial biological processes. Microenvironment Cav3.2 downregulated numerous genes associated with regulating the OPC cell state in GBM tumors such as SOX10 and Olig2. Neuronal Cav3.2 promoted neuron/GSC synaptic connections and GSC growth. Treatment of GSCs with the Cav3 blocker mibefradil downregulated genes associated with neuronal processes. The Cav3 blocker drug mibefradil synergized with temozolomide (TMZ) and radiation to reduce in vivo tumor growth and prolong animal survival.
Conclusions: Together these data reveal a role for microenvironment Cav3 in promoting GBM tumor progression through regulating neuronal and glial processes particularly associated with the OPC-cell state. Targeting both intrinsic and microenvironment Cav3 with the inhibitor mibefradil significantly enhanced the anti-GBM effects of TMZ and radiation.
Conflict of interest statement
Conflict of Interest The authors have declared that no conflicts of interest exist
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
