Efficacy and Safety of Bendamustine-Rituximab as Frontline Therapy for Indolent Non-Hodgkin Lymphoma: A Real-World, Single-Center, Retrospective Study
- PMID: 39229411
- PMCID: PMC11370709
- DOI: 10.7759/cureus.66124
Efficacy and Safety of Bendamustine-Rituximab as Frontline Therapy for Indolent Non-Hodgkin Lymphoma: A Real-World, Single-Center, Retrospective Study
Abstract
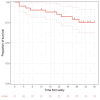
Background The use of bendamustine with an anti-CD20 monoclonal antibody as frontline therapy for indolent non-Hodgkin lymphoma (NHL) has become a standard of care. We aimed to evaluate the real-world efficacy and safety of bendamustine-rituximab (BR) frontline therapy for indolent NHL. Patients and methods Patients with indolent NHL treated with frontline BR therapy in Hôpital du Sacré-Coeur de Montréal, from January 2015 to August 2018 were included in this retrospective study. Results Our cohort included 42 adults with a median age of 63 years. Follicular lymphoma was the most common histology (n = 31, 74%). Most patients had advanced disease (Lugano stage III or IV, 88%). The overall response rate was 84% (complete response = 62% and partial response = 22%). Median progression-free survival (PFS) was not reached. At 30 months, PFS was 74.8% and overall survival was 90%. Grade 3-4 neutropenia occurred in 21% of patients. Infection-related adverse events were observed in 17 patients (40%). Most were grade 1 and 2 events (84%). One case of grade 5 progressive multifocal leukoencephalopathy related to John Cunningham (JC) virus reactivation was observed. The most common non-infectious-related adverse events were mild nausea and fatigue. Conclusions The efficacy and safety of BR treatment for indolent NHL were comparable in our real-life cohort compared to prior studies. This supports BR as a standard of care for indolent NHL. Future studies should assess whether the use of granulocyte-colony stimulating factors as primary prophylaxis effectively mitigates the hematological and infection-related adverse events related to BR therapy.
Keywords: bendamustine; frontline therapy; indolent lymphoma; treatment efficacy; treatment toxicities.
Copyright © 2024, Chan et al.
Conflict of interest statement
Human subjects: Consent was obtained or waived by all participants in this study. Comité d'éthique de la recherche du CIUSSS du Nord-de-l'Île-de-Montréal issued approval 2020-1931. Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue. Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: Jean-Samuel Boudreault: AbbVie, Janssen, Pfizer, and Sanofi. The other authors declare no conflict of interest.
Figures

References
-
- Comparison in low-tumor-burden follicular lymphomas between an initial no-treatment policy, prednimustine, or interferon alfa: a randomized study from the Groupe d'Etude des Lymphomes Folliculaires. Groupe d'Etude des Lymphomes de l'Adulte. Brice P, Bastion Y, Lepage E, et al. J Clin Oncol. 1997;15:1110–1117. - PubMed
-
- Current trends in diagnosis and management of follicular lymphoma. Gupta G, Garg V, Mallick S, Gogia A. https://pubmed.ncbi.nlm.nih.gov/36147608/ Am J Blood Res. 2022;12:105–124. - PMC - PubMed
-
- CVP chemotherapy plus rituximab compared with CVP as first-line treatment for advanced follicular lymphoma. Marcus R, Imrie K, Belch A, et al. Blood. 2005;105:1417–1423. - PubMed
LinkOut - more resources
Full Text Sources
