Molecular and biochemical components associated with chilling tolerance in tomato: comparison of different developmental stages
- PMID: 39232835
- PMCID: PMC11375913
- DOI: 10.1186/s43897-024-00108-0
Molecular and biochemical components associated with chilling tolerance in tomato: comparison of different developmental stages
Abstract
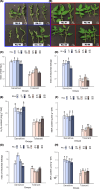
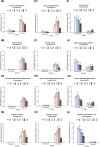
The cultivated tomato, Solanum lycopersicum, is highly sensitive to cold stress (CS), resulting in significant losses during cultivation and postharvest fruit storage. Previously, we demonstrated the presence of substantial genetic variation in fruit chilling tolerance in a tomato recombinant inbred line (RIL) population derived from a cross between a chilling-sensitive tomato line and a chilling-tolerant accession of the wild species S. pimpinellifolium. Here, we investigated molecular and biochemical components associated with chilling tolerance in fruit and leaves, using contrasting groups of "chilling tolerant" and "chilling sensitive" RI lines. Transcriptomic analyses were conducted on fruit exposed to CS, and gene expressions and biochemical components were measured in fruit and leaves. The analyses revealed core responding genes specific to either the cold-tolerant or cold-sensitive RI lines, which were differentially regulated in similar fashion in both leaves and fruit within each group. These genes may be used as markers to determine tomato germplasm cold tolerance or sensitivity. This study demonstrated that tomato response to CS in different developmental stages, including seedling and postharvest fruit, might be mediated by common biological/genetic factors. Therefore, genetic selection for cold tolerance during early stages of plant development may lead to lines with greater postharvest fruit chilling tolerance.
Keywords: Chilling; Cold; Postharvest; Storage; Tomato.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests. Dr. Amnon Lers is a member of the Editorial Board for Molecular Horticulture. He was not involved in the journal's review of, and decisions related to, this manuscript.
Figures




References
LinkOut - more resources
Full Text Sources
