A combination of Δ9-tetrahydrocannabinol and cannabidiol modulates glutamate dynamics in the hippocampus of an animal model of Alzheimer's disease
- PMID: 39232876
- PMCID: PMC11581878
- DOI: 10.1016/j.neurot.2024.e00439
A combination of Δ9-tetrahydrocannabinol and cannabidiol modulates glutamate dynamics in the hippocampus of an animal model of Alzheimer's disease
Abstract
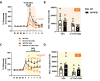
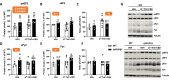
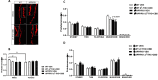
A combination of Δ9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD) at non-psychoactive doses was previously demonstrated to reduce cognitive decline in APP/PS1 mice, an animal model of Alzheimer's disease (AD). However, the neurobiological substrates underlying these therapeutic properties of Δ9-THC and CBD are not fully understood. Considering that dysregulation of glutamatergic activity contributes to cognitive impairment in AD, the present study evaluates the hypothesis that the combination of these two natural cannabinoids might reverse the alterations in glutamate dynamics within the hippocampus of this animal model of AD. Interestingly, our findings reveal that chronic treatment with Δ9-THC and CBD, but not with any of them alone, reduces extracellular glutamate levels and the basal excitability of the hippocampus in APP/PS1 mice. These effects are not related to significant changes in the function and structure of glutamate synapses, as no relevant changes in synaptic plasticity, glutamate signaling or in the levels of key components of these synapses were observed in cannabinoid-treated mice. Our data instead indicate that these cannabinoid effects are associated with the control of glutamate uptake and/or to the regulation of the hippocampal network. Taken together, these results support the potential therapeutic properties of combining these natural cannabinoids against the excitotoxicity that occurs in AD brains.
Keywords: Alzheimer; Cannabidiol; Cannabinoid; Glutamate; Hippocampus; Δ(9)-tetrahydrocannabinol.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Ester Aso reports financial support was provided by Spain Ministry of Science and Innovation. Jordi Bonaventura reports financial support was provided by Spain Ministry of Science and Innovation. Francisco Ciruela reports financial support was provided by Spain Ministry of Science and Innovation. Xavier Gasull reports financial support was provided by Spain Ministry of Science and Innovation. Francisco Ciruela reports financial support was provided by Departament de Recerca i Universitats de la Generalitat de Catalunya. Ester Aso reports financial support was provided by CYTED-Programa Iberoamericano de Ciencia y Tecnología para el Desarrollo. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures



References
-
- Alzheimer’s Association 2022 Alzheimer's disease facts and figures. Alzheimers Dement. 2022;18 - PubMed
-
- Howard R., McShane R., Lindesay J., Ritchie C., Baldwin A., Barber R., et al. Donepezil and memantine for moderate-to-severe Alzheimer's disease. N Engl J Med. 2012;366:893–903. - PubMed
-
- Liu K.Y., Walsh S., Brayne C., Merrick R., Richard E., Howard R. Evaluation of clinical benefits of treatments for Alzheimer's disease. Lancet Healthy Longev. 2023;4:e645–e651. - PubMed
-
- Solymosi K., Köfalvi A. Cannabis: a treasure trove or pandora's box? Mini Rev Med Chem. 2017;17:1223–1291. - PubMed

