Variable Craniofacial Shape and Development among Multiple Cave-Adapted Populations of Astyanax mexicanus
- PMID: 39234027
- PMCID: PMC11372417
- DOI: 10.1093/iob/obae030
Variable Craniofacial Shape and Development among Multiple Cave-Adapted Populations of Astyanax mexicanus
Abstract
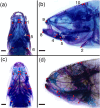
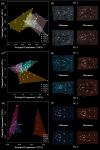
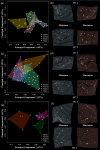
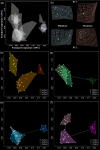
Astyanax mexicanus is a freshwater fish species with blind cave morphs and sighted surface morphs. Like other troglodytic species, independently evolved cave-dwelling A. mexicanus populations share several stereotypic phenotypes, including the expansion of certain sensory systems, as well as the loss of eyes and pigmentation. Here, we assess the extent to which there is also parallelism in craniofacial development across cave populations. Since multiple forces may be acting upon variation in the A. mexicanus system, including phylogenetic history, selection, and developmental constraint, several outcomes are possible. For example, eye regression may have triggered a conserved series of compensatory developmental events, in which case we would expect to observe highly similar craniofacial phenotypes across cave populations. Selection for cave-specific foraging may also lead to the evolution of a conserved craniofacial phenotype, especially in regions of the head directly associated with feeding. Alternatively, in the absence of a common axis of selection or strong developmental constraints, craniofacial shape may evolve under neutral processes such as gene flow, drift, and bottlenecking, in which case patterns of variation should reflect the evolutionary history of A. mexicanus. Our results found that cave-adapted populations do share certain anatomical features; however, they generally did not support the hypothesis of a conserved craniofacial phenotype across caves, as nearly every pairwise comparison was statistically significant, with greater effect sizes noted between more distantly related cave populations with little gene flow. A similar pattern was observed for developmental trajectories. We also found that morphological disparity was lower among all three cave populations versus surface fish, suggesting eye loss is not associated with increased variation, which would be consistent with a release of developmental constraint. Instead, this pattern reflects the relatively low genetic diversity within cave populations. Finally, magnitudes of craniofacial integration were found to be similar among all groups, meaning that coordinated development among anatomical units is robust to eye loss in A. mexicanus. We conclude that, in contrast to many conserved phenotypes across cave populations, global craniofacial shape is more variable, and patterns of shape variation are more in line with population structure than developmental architecture or selection.
Astyanax mexicanus es una especie de pez de agua dulce con morfologías de cueva ciega y morfologías de superficie videntes. Al igual que otras especies trogloditas, las poblaciones de A. mexicanus que habitan en cavernas evolucionaron independientemente y comparten varios fenotipos estereotipados. Entre ellos, la clave es la expansión de ciertos sistemas sensoriales y la pérdida de pigmentación y ojos. Aquí, evaluaremos hasta qué punto existen ciertos paralelismos en el desarrollo craneofacial entre poblaciones de cavernas conevolucion independientemente. Es posible que múltiples factores estén presentes actuando sobre la variación en el sistema de A. mexicanus, lo que lleva a varios posibles resultados. Por ejemplo, la regresión ocular se pudo haber desencadenado con una serie conservada de eventos de desarrollo compensatorio, en cuyo caso esperaríamos observar fenotipos craneofaciales muy similares en todas las poblaciones de cavernas. Además, dadas las demandas metabólicas y estructurales del ojo, su ausencia puede constituir una liberación de restricciones, lo que lleva a una expansión de la variaciónes craneofacial, la observación de diferentes patrones de covariación en relación con los peces de superficie, o ambos. Alternativamente, la selección para buscar alimento en cuevas específicas junto con la pérdida de ojos pudo haber desencadenado una línea evolutiva hacia un fenotipo craneofacial conservado. La variación intra e interpoblacional refleja en la historia evolutiva de A. mexicanus y en fuerzas demográficas como el flujo, la deriva y los cuellos de botella de genes. Nuestros resultados no respaldan un fenotipo craneofacial conservado en todas las cuevas, ya que casi todas las comparaciones por pares fueron estadísticamente significativas. Sin embargo, las poblaciones adaptadas a cuevas comparten ciertas características anatómicas. También observamos que la disparidad morfológica fue menor entre las tres poblaciones de cuevas en comparación con los peces de la superficie, lo que sugiere que la pérdida de ojos no está asociada con una mayor variación. En cambio, este patrón refleja la diversidad genética relativamente baja en las cuevas en comparación con las poblaciones de la superficie. Las magnitudes de la integración craneofacial también fueron similares entre todos los grupos, lo que significa que el desarrollo coordinado entre unidades anatómicas es resistente a la pérdida ocular en A. mexicanus. Concluimos que, a diferencia de muchos fenotipos conservados en las poblaciones de cuevas, la forma craneofacial global es más variable y los patrones de variación de la forma están más en línea con la estructura de la población que con el desarrollo arquitectural.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Society for Integrative and Comparative Biology.
Conflict of interest statement
The authors declare no competing interest.
Figures

Similar articles
-
Repeated evolution of eye loss in Mexican cavefish: Evidence of similar developmental mechanisms in independently evolved populations.J Exp Zool B Mol Dev Evol. 2020 Nov;334(7-8):423-437. doi: 10.1002/jez.b.22977. Epub 2020 Jul 2. J Exp Zool B Mol Dev Evol. 2020. PMID: 32614138
-
Phylogeographic relationships and morphological evolution between cave and surface Astyanax mexicanus populations (De Filippi 1853) (Actinopterygii, Characidae).Mol Ecol. 2023 Oct;32(20):5626-5644. doi: 10.1111/mec.17128. Epub 2023 Sep 15. Mol Ecol. 2023. PMID: 37712324
-
A pleiotropic interaction between vision loss and hypermelanism in Astyanax mexicanus cave x surface hybrids.BMC Evol Biol. 2016 Jun 30;16(1):145. doi: 10.1186/s12862-016-0716-y. BMC Evol Biol. 2016. PMID: 27363593 Free PMC article.
-
The complex origin of Astyanax cavefish.BMC Evol Biol. 2012 Jun 30;12:105. doi: 10.1186/1471-2148-12-105. BMC Evol Biol. 2012. PMID: 22747496 Free PMC article. Review.
-
Astyanax surface and cave fish morphs.Evodevo. 2020 Jul 11;11:14. doi: 10.1186/s13227-020-00159-6. eCollection 2020. Evodevo. 2020. PMID: 32676179 Free PMC article. Review.
References
-
- Adams D, Collyer ML, Kaliontzopoulou A. 2018. Geomorph: software for geometric morphometric analysis. R package version 3.0.6 (https://cran.r-project.org/package=geomorph).
-
- Aristotle . 1910. De Generatione Animalium by ArthurPlatt M.A.Professor of Greek in University College London Late Fellow of Trinity College Cambridge. Oxford, England: Clarendon Press.
LinkOut - more resources
Full Text Sources
