Vitamin D Alleviates Heavy Metal-Induced Cytotoxic Effects on Human Bone Osteoblasts Via the Induction of Bioenergetic Disruption, Oxidative Stress, and Apoptosis
- PMID: 39235540
- PMCID: PMC11920318
- DOI: 10.1007/s12011-024-04337-8
Vitamin D Alleviates Heavy Metal-Induced Cytotoxic Effects on Human Bone Osteoblasts Via the Induction of Bioenergetic Disruption, Oxidative Stress, and Apoptosis
Abstract
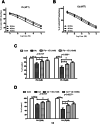
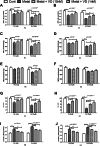
Cadmium (Cd) and lead (Pb) are heavy metals (HMs) that persistently contaminate the ecosystem, and bioaccumulation in bones is a health concern. We used biochemical and molecular assays to assess the cytoprotective effect of vitamin D (VD) on Cd- and Pd-induced chemical toxicity of human bone osteoblasts in vitro. Exposing Cd and Pb to human osteoblast cultures at concentrations of 0.1-1000 µM for 24-72 h significantly reduced osteoblast viability in an exposure time- and concentration-dependent manner. The cytotoxic effect of Cd on osteoblasts was more severe than Pb's, with 72-h exposure estimated half maximal effective concentration (EC50) of 8 and 12 µM, respectively, and VD (1 and 10 nM) alleviated cytotoxicity. Bioenergetics assays of ATP, mitochondrial membrane potential, and mitochondrial complex I and III activity showed that both Cd and Pb (1 and 10 µM) inhibited cellular bioenergetics after 72-h exposure. Cd and Pb increased lipid peroxidation and reactive oxygen species with reduced catalase/superoxide dismutase antioxidant activities and increased activity of caspases -3, -8, and -9. Co-treatment with VD (1 and 10 nM) counteracted bioenergetic disruption, oxidative damage, and apoptosis in a concentration-dependent manner. These findings suggest that VD is effective in managing the toxic effects of environmental pollutants and in treating bone diseases characterized by oxidative stress, apoptosis, and bioenergetic disruption.
Keywords: Antioxidants; Bone; Cadmium; Lead; Osteoblasts; Redox stress; Vitamin D.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing Interests: The authors declare no competing interests.
Figures




Similar articles
-
Fucoxanthin alleviates the cytotoxic effects of cadmium and lead on a human osteoblast cell line.Toxicol Res (Camb). 2024 Dec 19;13(6):tfae218. doi: 10.1093/toxres/tfae218. eCollection 2024 Dec. Toxicol Res (Camb). 2024. PMID: 39712643
-
The heavy metals lead and cadmium are cytotoxic to human bone osteoblasts via induction of redox stress.PLoS One. 2019 Nov 22;14(11):e0225341. doi: 10.1371/journal.pone.0225341. eCollection 2019. PLoS One. 2019. PMID: 31756223 Free PMC article.
-
Cadmium and lead induce mitochondrial dysfunction in ovarian theca Cells: Mechanisms of oxidative stress and bioenergetic collapse.Food Chem Toxicol. 2025 Aug;202:115531. doi: 10.1016/j.fct.2025.115531. Epub 2025 May 7. Food Chem Toxicol. 2025. PMID: 40345518
-
Advances in metal-induced oxidative stress and human disease.Toxicology. 2011 May 10;283(2-3):65-87. doi: 10.1016/j.tox.2011.03.001. Epub 2011 Mar 23. Toxicology. 2011. PMID: 21414382 Review.
-
Mitochondrial Oxidative Stress Is the General Reason for Apoptosis Induced by Different-Valence Heavy Metals in Cells and Mitochondria.Int J Mol Sci. 2023 Sep 22;24(19):14459. doi: 10.3390/ijms241914459. Int J Mol Sci. 2023. PMID: 37833908 Free PMC article. Review.
Cited by
-
The independent and interactive effects of heavy metal pollution and vitamin D deficiency on early kidney injury indicators: analysis of the National Health and Nutrition Examination Survey 2001-2004.BMC Public Health. 2025 Feb 21;25(1):719. doi: 10.1186/s12889-025-21796-3. BMC Public Health. 2025. PMID: 39984925 Free PMC article.
References
-
- Bharti R, Sharma R (2022) Effect of heavy metals: an overview. Mater Today: Proc 51:880–885
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

