Impact of CYP2C19 Genotype Status on Clinical Outcomes in Patients with Symptomatic Coronary Artery Disease, Stroke, and Peripheral Arterial Disease: A Systematic Review and Meta-Analysis
- PMID: 39235670
- PMCID: PMC11512840
- DOI: 10.1007/s40265-024-02076-7
Impact of CYP2C19 Genotype Status on Clinical Outcomes in Patients with Symptomatic Coronary Artery Disease, Stroke, and Peripheral Arterial Disease: A Systematic Review and Meta-Analysis
Abstract
Background: Clopidogrel is widely used for the secondary prevention of atherothrombotic events in patients with coronary artery disease (CAD), ischemic stroke, and peripheral arterial disease (PAD). CYP2C19 plays a pivotal role in the conversion of clopidogrel to its active metabolite. Clopidogrel-treated carriers of a CYP2C19 loss-of-function allele (LOF) may have a higher risk of new atherothrombotic events. Previous studies on genotype-guided treatment were mainly performed in CAD and showed mixed results.
Purpose: To simultaneously investigate the impact of CYP2C19 genotype status on the rate of atherothrombotic events in the most common types of atherosclerotic disease (CAD, stroke, PAD).
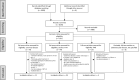
Methods: A comprehensive search in Pubmed, EMBASE, and MEDLINE from their inception to July 23rd 2023 was performed. Randomized controlled trials (RCTs) comparing genotype-guided and standard antithrombotic treatment, and cohort studies and post hoc analyses of RCTs concerning the association between CYP2C19 genotype status and clinical outcomes in clopidogrel-treated patients were included. The primary efficacy endpoint was major adverse cardiovascular events (MACE) and the safety end point major bleeding. Secondary endpoints were myocardial infarction, stent thrombosis, and ischemic stroke.
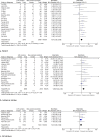
Results: Forty-four studies were identified: 11 studies on CAD, 29 studies on stroke, and 4 studies on PAD. In CAD, genotype-guided therapy significantly reduced the risk of MACE [risk ratio (RR) 0.60, 95% confidence interval (CI) 0.43-0.83], myocardial infarction (RR 0.53, 95% CI 0.42-0.68), and stent thrombosis (RR 0.64, 95% CI 0.43-0.94), compared with standard antithrombotic treatment. The rate of major bleeding did not differ significantly (RR 0.93, 95% CI 0.70-1.23). Most RCTs were performed in patients after percutaneous coronary intervention (9/11). In stroke, LOF carriers had a significantly higher risk of MACE (RR 1.61, 95% CI 1.25-2.08) and recurrent ischemic stroke (RR 1.89, 95% CI 1.48-2.40) compared with non-carriers. No significant differences were found in major bleeding (RR 0.90, 95% CI 0.43-1.89). In the 6955 patients with symptomatic PAD treated with clopidogrel in the EUCLID trial, no differences in MACE or major bleeding were found between LOF carriers and non-carriers. In three smaller studies on patients with PAD treated with clopidogrel after endovascular therapy, CYP2C19 genotype status was significantly associated with atherothrombotic events.
Conclusions: Genotype-guided treatment significantly decreased the rate of atherothrombotic events in patients with CAD, especially after PCI. In patients with history of stroke, LOF carriers treated with clopidogrel had a higher risk of MACE and recurrent stroke. The available evidence in PAD with regard to major adverse limb events is too limited to draw meaningful conclusions.
Registration: PROSPERO identifier no. CRD42020220284.
© 2024. The Author(s).
Conflict of interest statement
D.P.M.S.M.M., L.H.W., J.K., C.K., and M.C.W. declare that they have no potential conflicts of interest that might be relevant to the contents of this manuscript.
Figures




References
-
- Mega JL, Close SL, Wiviott SD, et al. Cytochrome p-450 polymorphisms and response to clopidogrel. N Engl J Med. 2009;360(4):354–62. - PubMed
-
- Sorich MJ, Rowland A, McKinnon RA, et al. CYP2C19 genotype has a greater effect on adverse cardiovascular outcomes following percutaneous coronary intervention and in Asian populations treated with clopidogrel: a meta-analysis. Circ Cardiovasc Genet. 2014;7(6):895–902. - PubMed
-
- Niu X, Mao L, Huang Y, et al. CYP2C19 polymorphism and clinical outcomes among patients of different races treated with clopidogrel: a systematic review and meta-analysis. J Huazhong Univ Sci Technol Med Sci. 2015;35(2):147–56. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

