Linked-evidence modelling of qualitative G6PD testing to inform low- and intermediate-dose primaquine treatment for radical cure of Plasmodium vivax
- PMID: 39236082
- PMCID: PMC11407642
- DOI: 10.1371/journal.pntd.0012486
Linked-evidence modelling of qualitative G6PD testing to inform low- and intermediate-dose primaquine treatment for radical cure of Plasmodium vivax
Abstract
Background: Radical cure of Plasmodium vivax infections is key to the control of vivax malaria. However, the standard doses of 8-aminoquinoline drugs used for radical cure can cause severe haemolysis in G6PD-deficient patients. The availability of near-patient G6PD tests could increase use of primaquine (PQ), however direct evidence of the impacts that G6PD testing has on downstream patient outcomes, such as haemolysis and recurrence is lacking.
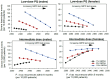
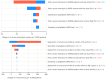
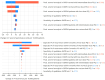
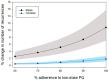
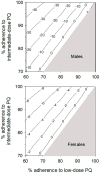
Methodology/principle findings: A linked-evidence model was created to investigate changes in the number of severe haemolysis events and P. vivax recurrences within 6 months of treatment when qualitative G6PD testing was used to guide PQ treatment (0.25mg/kg/day for 14 days and 0.5mg/kg/day for 7 days), compared to prescribing 14-day PQ with no G6PD testing. In the model patients identified as G6PD-deficient received 8-week PQ (0.75mg/kg/week). The model was used to simulate scenarios with 1%, 5% and 10% prevalence of G6PD-deficiency (G6PDd) in theoretical populations of 10,000 male and female P. vivax patients and initially assumed 100% adherence to the prescribed PQ regiment. Results illustrate that G6PD testing to guide the 14-day PQ regiment reduced severe haemolysis by 21-80% and increased recurrences by 3-6%, compared to applying the 14-day PQ regiment without G6PD testing. Results for the 7-day PQ regiment informed by G6PD testing were mixed, dependent on G6PDd prevalence and sex. When adherence to the PQ regiments was less than perfect the model predicted reductions in the number of recurrences at all prevalence levels, provided adherence to 7-day PQ was 5-10% higher than adherence to the 14-day regiment.
Conclusions/significance: Introduction of G6PD testing to guide PQ treatment reduces severe haemolysis events for the 14-day regiment, and the 7-day regiment in higher G6PDd prevalence settings, compared to use of 14-day PQ without G6PD testing when all patients adhere to the prescribed PQ treatment. At a population level, there were increases in recurrences, but this could be resolved when the 7-day regiment was used and had superior adherence compared to the 14-day regiment.
Copyright: © 2024 Michelle L. Gatton. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Primaquine alternative dosing schedules for preventing malaria relapse in people with Plasmodium vivax.Cochrane Database Syst Rev. 2020 Aug 19;8:CD012656. doi: 10.1002/14651858.CD012656.pub3. Cochrane Database Syst Rev. 2020. PMID: 32816320
-
G6PD deficiency, primaquine treatment, and risk of haemolysis in malaria-infected patients.Malar J. 2018 Nov 8;17(1):415. doi: 10.1186/s12936-018-2564-2. Malar J. 2018. PMID: 30409136 Free PMC article.
-
High daily dose Short COurse PrimaquinE after G6PD testing for the radical cure of Plasmodium vivax malaria in Indonesia and Papua New Guinea: the SCOPE implementation study protocol.BMC Infect Dis. 2025 Jul 16;25(1):922. doi: 10.1186/s12879-025-11109-9. BMC Infect Dis. 2025. PMID: 40670924 Free PMC article.
-
The risk of adverse clinical outcomes following treatment of Plasmodium vivax malaria with and without primaquine in Papua, Indonesia.PLoS Negl Trop Dis. 2020 Nov 11;14(11):e0008838. doi: 10.1371/journal.pntd.0008838. eCollection 2020 Nov. PLoS Negl Trop Dis. 2020. PMID: 33175835 Free PMC article.
-
Tafenoquine for preventing relapse in people with Plasmodium vivax malaria.Cochrane Database Syst Rev. 2020 Sep 6;9(9):CD010458. doi: 10.1002/14651858.CD010458.pub3. Cochrane Database Syst Rev. 2020. PMID: 32892362 Free PMC article.
References
-
- World Health Organiazation. World Malaria Report 2023. Geneva; 2023. https://www.who.int/teams/global-malaria-programme/reports/world-malaria....
-
- Recht J, Ashley E, White N. Safety of 8-aminoquinoline antimalarial medicines. Geneva: World Health Organization; 2014.
-
- World Health Organiazation. WHO Guidelines for Malaria. Geneva; 2023 16 October 2023. Contract No.: WHO/UCN/GMP/2023.01 Rev. 1. https://www.who.int/teams/global-malaria-programme.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

