Antiviral effect of Evusheld in COVID-19 hospitalized patients infected with pre-Omicron or Omicron variants: a modelling analysis of the randomized DisCoVeRy trial
- PMID: 39236218
- PMCID: PMC11531825
- DOI: 10.1093/jac/dkae301
Antiviral effect of Evusheld in COVID-19 hospitalized patients infected with pre-Omicron or Omicron variants: a modelling analysis of the randomized DisCoVeRy trial
Abstract
Background: The antiviral efficacy of Evusheld (AZD7442) in patients hospitalized for SARS-CoV-2 is unknown.
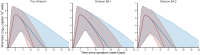
Methods: We analysed the evolution of both the nasopharyngeal viral load and the serum neutralization activity against the variant of infection in 199 hospitalized patients (109 treated with Evusheld, 90 treated with placebo) infected with the SARS-CoV-2 virus and included in the randomized, double-blind, trial DisCoVeRy (NCT04315948). Using a mechanistic mathematical model, we reconstructed the trajectories of viral kinetics and how they are modulated by the increase in serum neutralization activity during Evusheld treatment.
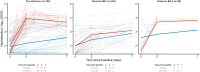
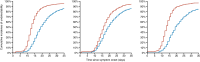
Results: Our model identified that the neutralization activity was associated with viral kinetics. Reflecting the variant-dependent neutralization activity of Evusheld, the antiviral activity of Evusheld was larger in patients infected with pre-Omicron or Omicron BA.2 variants than in patients infected with Omicron BA.1 variant. More specifically, the model predicted that Evusheld reduced the median time to viral clearance compared with placebo-treated patients by more than 5 days in patients infected by pre-Omicron (median: 5.9; 80% PI: 2.1-13.6) or Omicron BA.2 (median: 5.4; 80% PI: 2.0-12.4), respectively. The effect was more modest in patients infected by the Omicron BA.1 variant, reducing the median time to viral clearance by 2 days (median: 2.2; 80% PI: 0.4-8.9).
Conclusions: Hospitalized patients treated with Evusheld had a shorter median time to SARS-CoV-2 viral clearance. As Evusheld antiviral activity is mediated by the level of neutralization activity, its impact on viral clearance varies largely according to the variant of infection.
© The Author(s) 2024. Published by Oxford University Press on behalf of British Society for Antimicrobial Chemotherapy.
Figures




Comment in
-
Comment on: Antiviral effect of Evusheld in COVID-19 hospitalized patients infected with pre-Omicron or Omicron variants: a modelling analysis of the randomized DisCoVeRy trial.J Antimicrob Chemother. 2025 Mar 3;80(3):891-892. doi: 10.1093/jac/dkae385. J Antimicrob Chemother. 2025. PMID: 39499209 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Supplementary concepts
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

