Glioblastoma mesenchymal subtype enhances antioxidant defence to reduce susceptibility to ferroptosis
- PMID: 39237744
- PMCID: PMC11377710
- DOI: 10.1038/s41598-024-72024-8
Glioblastoma mesenchymal subtype enhances antioxidant defence to reduce susceptibility to ferroptosis
Abstract
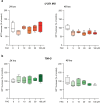
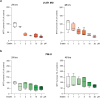
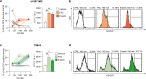
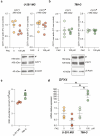
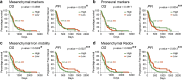
Glioblastoma (GBM) represents an aggressive brain tumor, characterized by intra- and inter-tumoral heterogeneity and therapy resistance, leading to unfavourable prognosis. An increasing number of studies pays attention on the regulation of ferroptosis, an iron-dependent cell death, as a strategy to reverse drug resistance in cancer. However, the debate on whether this strategy may have important implications for the treatment of GBM is still ongoing. In the present study, we used ferric ammonium citrate and erastin to evaluate ferroptosis induction effects on two human GBM cell lines, U-251 MG, with proneural characteristics, and T98-G, with a mesenchymal profile. The response to ferroptosis induction was markedly different between cell lines, indeed T98-G cells showed an enhanced antioxidant defence, with increased glutathione levels, as compared to U-251 MG cells. Moreover, using bioinformatic approaches and analysing publicly available datasets from patients' biopsies, we found that GBM with a mesenchymal phenotype showed an up-regulation of several genes involved in antioxidant mechanisms as compared to proneural subtype. Thus, our results suggest that GBM subtypes differently respond to ferroptosis induction, emphasizing the significance of further molecular studies on GBM to better discriminate between various tumor subtypes and progressively move towards personalized therapy.
Keywords: Erastin; Ferroptosis; Glioblastoma; Iron overload; Mesenchymal subtype; Proneural subtype.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
MeSH terms
Substances
Grants and funding
- PNC0000003/National Plan for NRRP Complementary Investments (PNC, established with the decree-law 6 May 2021, n. 59, converted by law n. 101 of 2021) in the call for the funding of research initiatives for technologies and innovative trajectories in the health and care sectors (Directorial Decree n. 931 of 06-06-2022) - AdvaNced Technologies for Human-centrEd Medicine (project acronym: ANTHEM)
- CHRONOS/Piano di Incentivi per la Ricerca di Ateneo 2020-2022, Linea di intervento 3-Starting Grant
- PON03PE_00216_1/Drug Delivery: veicoli per un'innovazione sostenibile" (CUP: B82F15000010005. Funder "Ministry of University and Research", ITALY)
- MD-RESETT-GLIO/Piano di Incentivi per la Ricerca di Ateneo 2020-2022, Linea di Intervento 2
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

