Orally biomimetic metal-phenolic nanozyme with quadruple safeguards for intestinal homeostasis to ameliorate ulcerative colitis
- PMID: 39238009
- PMCID: PMC11378530
- DOI: 10.1186/s12951-024-02802-z
Orally biomimetic metal-phenolic nanozyme with quadruple safeguards for intestinal homeostasis to ameliorate ulcerative colitis
Abstract
Background: Ulcerative colitis (UC) is defined by persistent inflammatory processes within the gastrointestinal tract of uncertain etiology. Current therapeutic approaches are limited in their ability to address oxidative stress, inflammation, barrier function restoration, and modulation of gut microbiota in a coordinated manner to maintain intestinal homeostasis.
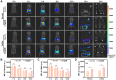
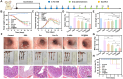
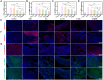
Results: This study involves the construction of a metal-phenolic nanozyme (Cur-Fe) through a ferric ion-mediated oxidative coupling of curcumin. Cur-Fe nanozyme exhibits superoxide dismutase (SOD)-like and •OH scavenging activities, demonstrating significant anti-inflammatory and anti-oxidant properties for maintaining intracellular redox balance in vitro. Drawing inspiration from Escherichia coli Nissle 1917 (EcN), a biomimetic Cur-Fe nanozyme (CF@EM) is subsequently developed by integrating Cur-Fe into the EcN membrane (EM) to improve the in vivo targeting ability and therapeutic effectiveness of the Cur-Fe nanozyme. When orally administered, CF@EM demonstrates a strong ability to colonize the inflamed colon and restore intestinal redox balance and barrier function in DSS-induced colitis models. Importantly, CF@EM influences the gut microbiome towards a beneficial state by enhancing bacterial diversity and shifting the compositional structure toward an anti-inflammatory phenotype. Furthermore, analysis of intestinal microbial metabolites supports the notion that the therapeutic efficacy of CF@EM is closely associated with bile acid metabolism.
Conclusion: Inspired by gut microbes, we have successfully synthesized a biomimetic Cur-Fe nanozyme with the ability to inhibit inflammation and restore intestinal homeostasis. Collectively, without appreciable systemic toxicity, this work provides an unprecedented opportunity for targeted oral nanomedicine in the treatment of ulcerative colitis.
Keywords: Inflammation; Intestinal homeostasis; Metal-phenolic nanozyme; Oxidative stress; Ulcerative colitis.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






References
-
- Liu JZ, van Sommeren S, Huang H, Ng SC, Alberts R, Takahashi A, Ripke S, Lee JC, Jostins L, Shah T, et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat Genet. 2015;47:979–86. 10.1038/ng.3359 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

