Preventing Postpartum Venous Thromboembolism With Low-Molecular-Weight Heparin: The PP-HEP Pilot Randomised Controlled Trial
- PMID: 39238110
- PMCID: PMC11612613
- DOI: 10.1111/1471-0528.17943
Preventing Postpartum Venous Thromboembolism With Low-Molecular-Weight Heparin: The PP-HEP Pilot Randomised Controlled Trial
Abstract
Objective: Uncertainty surrounds the risk-benefit of low-molecular-weight heparin to prevent postpartum venous thromboembolism (VTE). Data from randomised clinical trials (RCT) are critically needed, but recent feasibility studies in North America yielded low participation rates, with <1 enrolment per month per centre. Our aim was to assess the feasibility of a trial of postpartum short-term enoxaparin in Europe.
Design: Pragmatic, open-label pilot randomised controlled trial (RCT).
Setting: Swiss tertiary hospital.
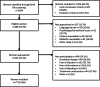
Population: Postpartum women, within 48 h of delivery, deemed at intermediate risk of VTE with at least one major risk factor (morbid obesity, thrombophilia, emergency caesarean section, pre-eclampsia, preterm delivery, intrauterine growth restriction or systemic peripartum infection) and/or at least two minor risk factors.
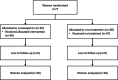
Methods: Participants were randomised to enoxaparin 40-60 mg once daily for 10 days or no treatment, with a 90-day follow-up.
Main outcome measures: Participation rate and study acceptance (randomised participants among women in whom informed consent was sought).
Results: Recruitment was open for 25 weeks in 2022. Among 1504 postpartum women, 480 were eligible and 77 were randomised. The recruitment rate was 3.1 per week (13.3 per month) and the study acceptance was 23.8%. At 3 months, there was no VTE event, but one major, one nonmajor obstetrical bleeding and one surgical site complication, all in the enoxaparin group.
Conclusions: This pilot RCT of postpartum thromboprophylaxis set in Switzerland yielded greater participation rate and acceptance than previous attempts in North America. It calls for a large, international, collaborative trial to guide this important clinical decision.
Trial registration: ClinicalTrial.gov identifier: NCT05878899 and NCT04153760.
Keywords: low‐molecular‐weight heparin; postpartum; pregnancy; venous thrombosis.
© 2024 The Author(s). BJOG: An International Journal of Obstetrics and Gynaecology published by John Wiley & Sons Ltd.
Conflict of interest statement
M.B. reports travel support from SOBI; B.M.D.T. reports consulting fees from Effik and Pierre‐Favre, honoraria for lectures from Effik and receipt of medical equipment from Pregnolia and Hologic; all other authors report no conflicts of interest.
Figures
References
-
- Roach R. E., Lijfering W. M., Rosendaal F. R., Cannegieter S. C., and le Cessie S., “Sex Difference in Risk of Second But Not of First Venous Thrombosis: Paradox Explained,” Circulation 129, no. 1 (2014): 51–56. - PubMed
-
- Wik H. S., Enden T. R., Jacobsen A. F., and Sandset P. M., “Long‐Term Quality of Life After Pregnancy‐Related Deep Vein Thrombosis and the Influence of Socioeconomic Factors and Comorbidity,” Journal of Thrombosis and Haemostasis 9, no. 10 (2011): 1931–1936. - PubMed
-
- Gassmann N., Viviano M., Righini M., Fontana P., Martinez de Tejada B., and Blondon M., “Estimating the Risk Thresholds Used by Guidelines to Recommend Postpartum Thromboprophylaxis,” Journal of Thrombosis and Haemostasis 19, no. 2 (2021): 452–459. - PubMed
-
- Merriam A. A., Huang Y., Ananth C. V., Wright J. D., D'Alton M. E., and Friedman A. M., “Postpartum Thromboembolism Prophylaxis During Delivery Hospitalizations,” American Journal of Perinatology 35, no. 9 (2018): 873–881. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous