Activation of Toll-like receptor 2 promotes mesenchymal stem/stromal cell-mediated immunoregulation and angiostasis through AKR1C1
- PMID: 39239520
- PMCID: PMC11373616
- DOI: 10.7150/thno.100327
Activation of Toll-like receptor 2 promotes mesenchymal stem/stromal cell-mediated immunoregulation and angiostasis through AKR1C1
Abstract
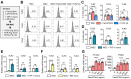
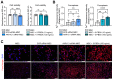
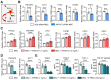
Background: Mesenchymal stem/stromal cells (MSCs) maintain tissue homeostasis in response to microenvironmental perturbations. Toll-like receptors (TLRs) are key sensors for exogenous and endogenous signals produced during injury. In this study, we aimed to investigate whether TLRs affect the homeostatic functions of MSCs after injury. Methods: We examined the expression of TLR2, TLR3 and TLR4 in MSCs, and analyzed the functional significance of TLR2 activation using single-cell RNA sequencing. Additionally, we investigated the effects and mechanisms of TLR2 and its downstream activation in MSCs on the MSCs themselves, on monocytes/macrophages, and in a mouse model of sterile injury-induced inflammatory corneal angiogenesis. Results: MSCs expressed TLR2, which was upregulated by monocytes/macrophages. Activation of TLR2 in MSCs promoted their immunoregulatory and angiostatic functions in monocytes/macrophages and in mice with inflammatory corneal angiogenesis, whereas TLR2 inhibition attenuated these functions. Single-cell RNA sequencing revealed AKR1C1, a gene encoding aldo-keto reductase family 1 member C1, as the most significantly inducible gene in MSCs upon TLR2 stimulation, though its stimulation did not affect cell compositions. AKR1C1 protected MSCs against ferroptosis, increased secretion of anti-inflammatory cytokines, and enhanced their ability to drive monocytes/macrophages towards immunoregulatory phenotypes, leading to the amelioration of inflammatory corneal neovascularization in mice. Conclusion: Our findings suggest that activation of TLR2-AKR1C1 signaling in MSCs serves as an important pathway for the survival and homeostatic activities of MSCs during injury.
Keywords: Ado-keto reductase family 1 member C1; Cornea; Mesenchymal stem/stromal cell; Monocyte/macrophage; Toll-like receptor 2.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures





References
-
- Cheung TS, Dazzi F. Mesenchymal-myeloid interaction in the regulation of immunity. Semin Immunol. 2018;35:59–68. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

