Paeoniflorin protects hepatocytes from APAP-induced damage through launching autophagy via the MAPK/mTOR signaling pathway
- PMID: 39244559
- PMCID: PMC11380789
- DOI: 10.1186/s11658-024-00631-4
Paeoniflorin protects hepatocytes from APAP-induced damage through launching autophagy via the MAPK/mTOR signaling pathway
Abstract
Background: Drug-induced liver injury (DILI) is gradually becoming a common global problem that causes acute liver failure, especially in acute hepatic damage caused by acetaminophen (APAP). Paeoniflorin (PF) has a wide range of therapeutic effects to alleviate a variety of hepatic diseases. However, the relationship between them is still poorly investigated in current studies.
Purpose: This work aimed to explore the protective effects of PF on APAP-induced hepatic damage and researched the potential molecular mechanisms.
Methods: C57BL/6J male mice were injected with APAP to establish DILI model and were given PF for five consecutive days for treatment. Aiming to clarify the pharmacological effects, the molecular mechanisms of PF in APAP-induced DILI was elucidated by high-throughput and other techniques.
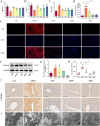
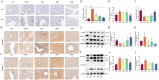
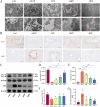
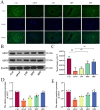
Results: The results demonstrated that serum levels of ALP, γ-GT, AST, TBIL, and ALT were decreased in APAP mice by the preventive effects of PF. Moreover, PF notably alleviated hepatic tissue inflammation and edema. Meanwhile, the results of TUNEL staining and related apoptotic factors coincided with the results of transcriptomics, suggesting that PF inhibited hepatocyte apoptosis by regulated MAPK signaling. Besides, PF also acted on reactive oxygen species (ROS) to regulate the oxidative stress for recovery the damaged mitochondria. More importantly, transmission electron microscopy showed the generation of autophagosomes after PF treatment, and PF was also downregulated mTOR and upregulated the expression of autophagy markers such as ATG5, ATG7, and BECN1 at the mRNA level and LC3, p62, ATG5, and ATG7 at the protein level, implying that the process by which PF exerted its effects was accompanied by the occurrence of autophagy. In addition, combinined with molecular dynamics simulations and western blotting of MAPK, the results suggested p38 as a direct target for PF on APAP. Specifically, PF-activated autophagy through the downregulation of MAPK/mTOR signaling, which in turn reduced APAP injury.
Conclusions: Paeoniflorin mitigated liver injury by activating autophagy to suppress oxidative stress and apoptosis via the MAPK/mTOR signaling pathway. Taken together, our findings elucidate the role and mechanism of paeoniflorin in DILI, which is expected to provide a new therapeutic strategy for the development of paeoniflorin.
Keywords: Acetaminophen; Cell death; Drug-induced liver injury; Natural products; Oxidative stress; Signal transduction.
© 2024. The Author(s).
Conflict of interest statement
All the authors declare that there have no conflicts of financial interests or publication in this work.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

