Mir221- and Mir222-enriched adsc-exosomes mitigate PM exposure-exacerbated cardiac ischemia-reperfusion injury through the modulation of the BNIP3-MAP1LC3B-BBC3/PUMA pathway
- PMID: 39245438
- PMCID: PMC11760231
- DOI: 10.1080/15548627.2024.2395799
Mir221- and Mir222-enriched adsc-exosomes mitigate PM exposure-exacerbated cardiac ischemia-reperfusion injury through the modulation of the BNIP3-MAP1LC3B-BBC3/PUMA pathway
Abstract
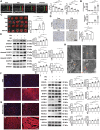
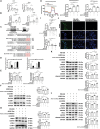
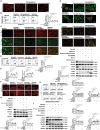
Epidemiology has shown a strong relationship between fine particulate matter (PM) exposure and cardiovascular disease. However, it remains unknown whether PM aggravates myocardial ischemia-reperfusion (I/R) injury, and the related mechanisms are unclear. Our previous study has shown that adipose stem cell-derived exosomes (ADSC-Exos) contain high levels of Mir221 and Mir222. The present study investigated the effects of PM exposure on I/R-induced cardiac injury through mitophagy and apoptosis, as well as the potential role of Mir221 and Mir222 in ADSC-Exos. Wild-type, mir221- and mir222-knockout (KO), and Mir221- and Mir222-overexpressing transgenic (TG) mice were intratracheally injected with PM (10 mg/kg). After 24 h, mice underwent left coronary artery ligation for 30 min, followed by 3 h of reperfusion (I/R). H9c2 cardiomyocytes were cultured under 1% O2 for 6 h, then reoxygenated for 12 h (hypoxia-reoxygenation [H/R]). PM aggravated I/R (or H/R) cardiac injury by increasing ROS levels and causing mitochondrial dysfunction, which increased the expression of mitochondrial fission-related proteins (DNM1L/Drp1 and MFF) and mitophagy-related proteins (BNIP3 and MAP1LC3B/LC3B) in vivo and in vitro. Treatment with ADSC-Exos or Mir221- and Mir222-mimics significantly reduced PM+I/R-induced cardiac injury. Importantly, ADSC-Exos contain Mir221 and Mir222, which directly targets BNIP3, MAP1LC3B/LC3B, and BBC3/PUMA, decreasing their expression and ultimately reducing cardiomyocyte mitophagy and apoptosis. The present data showed that ADSC-Exos treatment regulated mitophagy and apoptosis through the Mir221 and Mir222-BNIP3-MAP1LC3B-BBC3/PUMA pathway and significantly reduced the cardiac damage caused by PM+I/R. The present study revealed the novel therapeutic potential of ADSC-Exos in alleviating PM-induced exacerbation of myocardial I/R injury.Abbreviation: ADSC-Exos: adipose-derived stem cell exosomes; AL: autolysosome; ATP: adenosine triphosphate; BBC3/PUMA: BCL2 binding component 3; BNIP3: BCL2/adenovirus E1B interacting protein 3; CASP3: caspase 3; CASP9: caspase 9; CDKN1B/p27: cyclin dependent kinase inhibitor 1B; CVD: cardiovascular disease; DCFH-DA: 2',7'-dichlorodihydrofluorescein diacetate; DHE: dihydroethidium; DNM1L/Drp1: dynamin 1-like; EF: ejection fraction; FS: fractional shortening; H/R: hypoxia-reoxygenation; I/R: ischemia-reperfusion; LDH: lactate dehydrogenase; MAP1LC3B/LC3B: microtubule-associated protein 1 light chain 3 beta; MFF: mitochondrial fission factor; miRNA: microRNA; NAC: N-acetylcysteine; OCR: oxygen consumption rate; PIK3C3/Vps34: phosphatidylinositol 3-kinase catalytic subunit type 3; PM: particulate matter; PRKAA1/AMPK: protein kinase AMP-activated catalytic subunit alpha 1; ROS: reactive oxygen species; SQSTM1/p62: sequestosome 1; TEM: transmission electron microscopy; TRP53/p53: transformation related protein 53; TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling.
Keywords: ADSC-exosomes; Mir221 and Mir222; cardiomyocyte; ischemia/reperfusion injury; mitophagy; particulate matter.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
