Amygdala atrophies in specific subnuclei in preclinical Alzheimer's disease
- PMID: 39254209
- PMCID: PMC11485073
- DOI: 10.1002/alz.14235
Amygdala atrophies in specific subnuclei in preclinical Alzheimer's disease
Abstract
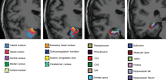
Introduction: Magnetic resonance imaging (MRI) segmentation algorithms make it possible to study detailed medial temporal lobe (MTL) substructures as hippocampal subfields and amygdala subnuclei, offering opportunities to develop biomarkers for preclinical Alzheimer's disease (AD).
Methods: We identified the MTL substructures significantly associated with tau-positron emission tomography (PET) signal in 581 non-demented individuals from the Alzheimer's Disease Neuroimaging Initiative (ADNI-3). We confirmed our results in our UCLouvain cohort including 110 non-demented individuals by comparing volumes between individuals with different visual Braak's stages and clinical diagnosis.
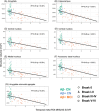
Results: Four amygdala subnuclei (cortical, central, medial, and accessory basal) were associated with tau in amyloid beta-positive (Aβ+) clinically normal (CN) individuals, while the global amygdala and hippocampal volumes were not. Using UCLouvain data, we observed that both Braak I-II and Aβ+ CN individuals had smaller volumes in these subnuclei, while no significant difference was observed in the global structure volumes or other subfields.
Conclusion: Measuring specific amygdala subnuclei, early atrophy may serve as a marker of temporal tauopathy in preclinical AD, identifying individuals at risk of progression.
Highlights: Amygdala atrophy is not homogeneous in preclinical Alzheimer's disease (AD). Tau pathology is associated with atrophy of specific amygdala subnuclei, specifically, the central, medial, cortical, and accessory basal subnuclei. Hippocampal and amygdala volume is not associated with tau in preclinical AD. Hippocampus and CA1-3 volume is reduced in preclinical AD, regardless of tau.
Keywords: AV1451; Alzheimer's disease; MK6240; amygdala subnuclei; biomarker; florquinitau; flortaucipir; preclinical AD; structural magnetic resonance imaging; tauopathy; tau‐positron emission tomography.
© 2024 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
The firm Lantheus Inc. supplied the [18F]MK6240 precursor for acquiring the PET images analyzed in this article. No other conflicts of interest are reported. Author disclosures are available in the supporting information.
Figures




References
-
- McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging‐Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7(3):263‐269. doi:10.1016/j.jalz.2011.03.005 - DOI - PMC - PubMed
-
- Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging‐Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7(3):280‐292. doi:10.1016/j.jalz.2011.03.003 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
- Laboratory for Neuro Imaging at the University of Southern California
- BioClinica, Inc
- Araclon Biotech
- GE Healthcare
- 40010035/FRFS-WELBIO
- Cogstate
- F. Hoffmann-La Roche Ltd
- Eli Lilly and Company
- Merck & Co., Inc.
- W81XWH-12-2-0012/Alzheimer's Disease Neuroimaging Initiative (ADNI)
- Neurotrack Technologies
- Eisai Inc.
- FRIA40014635/Belgian Fund for Scientific Research (FNRS)
- CCL40010417/Belgian Fund for Scientific Research (FNRS)
- EuroImmun
- Biogen
- CAPMC/ CIHR/Canada
- CereSpir, Inc
- Alzheimer's Drug Discovery Foundation
- Servier
- Lumosity
- Bristol-Myers Squibb Company
- U01 AG024904/AG/NIA NIH HHS/United States
- Alzheimer's Therapeutic Research Institute at the University of Southern California
- Piramal Imaging
- Takeda Pharmaceutical Company
- ALZ/Alzheimer's Association/United States
- Genentech, Inc.
- Novartis Pharmaceuticals Corporation
- Meso Scale Diagnostics, LLC.
- Northern California Institute for Research and Education
- Janssen Alzheimer Immunotherapy Research & Development, LLC.
- AbbVie
- IXICO Ltd
- Transition Therapeutics
- Belgian Alzheimer Research Foundation
- EB/NIBIB NIH HHS/United States
- Johnson & Johnson Pharmaceutical Research & Development LLC
- NH/NIH HHS/United States
- Pfizer Inc.
- Elan Pharmaceuticals, Inc.
- Department of Defense
- NeuroRx Research
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

