Synthesis and Optical Characterization of Hydrazone-Substituted Push-Pull-Type NLOphores
- PMID: 39255504
- PMCID: PMC11421011
- DOI: 10.1021/acs.joc.4c01328
Synthesis and Optical Characterization of Hydrazone-Substituted Push-Pull-Type NLOphores
Abstract
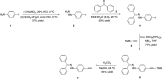
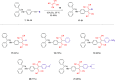
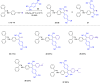
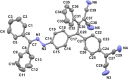
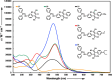
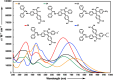
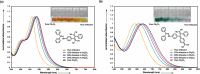
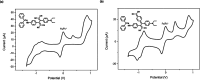
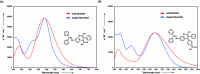
Two distinct families of NLOphores featuring hydrazone donors were synthesized using click-type [2 + 2] cycloaddition retroelectrocyclizations (CA-RE). Despite the limitations in the substrate scope, it was shown for the first time that hydrazone-activated alkynes could undergo reactions with TCNE/TCNQ. The electrochemical, photophysical, and second-order nonlinear optical (NLO) characteristics of the chromophores were analyzed utilizing experimental and computational approaches. Chromophores 17-21 and 23-27 exhibited two reduction waves, along with one oxidation wave that can be attributed to the hydrazone moiety. All chromophores exhibit charge-transfer bands extending from the visible to the near-infrared region. The λmax of hydrazone-based chromophores falls within the range of 473 to 725 nm. Additionally, all chromophores exhibited positive solvatochromism. Computational studies have been performed to elucidate the origin of the low-energy absorption bands. Parameters such as dipole moment, band gaps, electronegativity, global chemical hardness/softness, average polarizability, and first hyperpolarizability were calculated to obtain information about NLO properties of the target structures. The thermal stabilities of the NLOphores were assessed through TGA. Experimental NLO measurements were conducted using the electric field-induced second harmonic generation (EFISHG) technique. The studied structures demonstrated NLO responses, with μβ values between 520 × 10-48 esu and 5300 × 10-48 esu.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
Carbamate-Functionalized NLOphores via a Formal [2+2] Cycloaddition-Retroelectrocyclization Strategy.Chemistry. 2025 Mar 3;31(13):e202404778. doi: 10.1002/chem.202404778. Epub 2025 Jan 31. Chemistry. 2025. PMID: 39844739 Free PMC article.
-
Polycyclic aromatic hydrocarbon-substituted push-pull chromophores: an investigation of optoelectronic and nonlinear optical properties using experimental and theoretical approaches.Turk J Chem. 2021 Oct 19;45(5):1375-1390. doi: 10.3906/kim-2102-22. eCollection 2021. Turk J Chem. 2021. PMID: 34849054 Free PMC article.
-
BODIPY-bridged push-pull chromophores for nonlinear optical applications.Chemphyschem. 2014 Sep 15;15(13):2693-700. doi: 10.1002/cphc.201402123. Epub 2014 Jun 20. Chemphyschem. 2014. PMID: 24954812
-
The [2+2] Cycloaddition-Retroelectrocyclization (CA-RE) Click Reaction: Facile Access to Molecular and Polymeric Push-Pull Chromophores.Angew Chem Int Ed Engl. 2018 Mar 26;57(14):3552-3577. doi: 10.1002/anie.201711605. Epub 2018 Feb 22. Angew Chem Int Ed Engl. 2018. PMID: 29469183 Review.
-
Second-order nonlinear polarizability of "Push-Pull" chromophores. A decade of progress in donor-π-acceptor materials.Chem Rec. 2022 Jun;22(6):e202200024. doi: 10.1002/tcr.202200024. Epub 2022 Mar 29. Chem Rec. 2022. PMID: 35352466 Review.
Cited by
-
Carbamate-Functionalized NLOphores via a Formal [2+2] Cycloaddition-Retroelectrocyclization Strategy.Chemistry. 2025 Mar 3;31(13):e202404778. doi: 10.1002/chem.202404778. Epub 2025 Jan 31. Chemistry. 2025. PMID: 39844739 Free PMC article.
-
Orthogonal Functionalization of Oxo-Graphene Nanoribbons.Chemistry. 2025 Jan 22;31(5):e202403645. doi: 10.1002/chem.202403645. Epub 2024 Dec 2. Chemistry. 2025. PMID: 39556476 Free PMC article.
References
-
- Abolhasani M.; Kumacheva E. The Rise of Self-Driving Labs in Chemical and Materials Sciences. Nat. Synth. 2023, 2, 483–492. 10.1038/s44160-022-00231-0. - DOI
LinkOut - more resources
Full Text Sources

