Hypoxia and Foxn1 alter the proteomic signature of dermal fibroblasts to redirect scarless wound healing to scar-forming skin wound healing in Foxn1-/- mice
- PMID: 39256768
- PMCID: PMC11389453
- DOI: 10.1186/s12915-024-01990-2
Hypoxia and Foxn1 alter the proteomic signature of dermal fibroblasts to redirect scarless wound healing to scar-forming skin wound healing in Foxn1-/- mice
Abstract
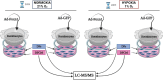
Background: Foxn1-/- deficient mice are a rare model of regenerative skin wound healing among mammals. In wounded skin, the transcription factor Foxn1 interacting with hypoxia-regulated factors affects re-epithelialization, epithelial-mesenchymal transition (EMT) and dermal white adipose tissue (dWAT) reestablishment and is thus a factor regulating scar-forming/reparative healing. Here, we hypothesized that transcriptional crosstalk between Foxn1 and Hif-1α controls the switch from scarless (regenerative) to scar-present (reparative) skin wound healing. To verify this hypothesis, we examined (i) the effect of hypoxia/normoxia and Foxn1 signalling on the proteomic signature of Foxn1-/- (regenerative) dermal fibroblasts (DFs) and then (ii) explored the effect of Hif-1α or Foxn1/Hif-1α introduced by a lentiviral (LV) delivery vector to injured skin of regenerative Foxn1-/- mice with particular attention to the remodelling phase of healing.
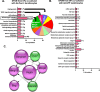
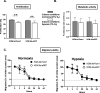
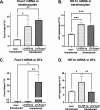
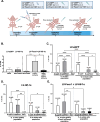
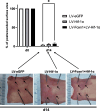
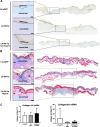
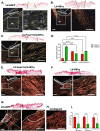
Results: We showed that hypoxic conditions and Foxn1 stimulation modified the proteome of Foxn1-/- DFs. Hypoxic conditions upregulated DF protein profiles, particularly those related to extracellular matrix (ECM) composition: plasminogen activator inhibitor-1 (Pai-1), Sdc4, Plod2, Plod1, Lox, Loxl2, Itga2, Vldlr, Ftl1, Vegfa, Hmox1, Fth1, and F3. We found that Pai-1 was stimulated by hypoxic conditions in regenerative Foxn1-/- DFs but was released by DFs to the culture media exclusively upon hypoxia and Foxn1 stimulation. We also found higher levels of Pai-1 protein in DFs isolated from Foxn1+/+ mice (reparative/scar-forming) than in DFs isolated from Foxn1-/- (regenerative/scarless) mice and triggered by injury increase in Foxn1 and Pai-1 protein in the skin of mice with active Foxn1 (Foxn1+/+ mice). Then, we demonstrated that the introduction of Foxn1 and Hif-1α via lentiviral injection into the wounded skin of regenerative Foxn1-/- mice activates reparative/scar-forming healing by increasing the wounded skin area and decreasing hyaluronic acid deposition and the collagen type III to I ratio. We also identified a stimulatory effect of LV-Foxn1 + LV-Hif-1α injection in the wounded skin of Foxn1-/- mice on Pai-1 protein levels.
Conclusions: The present data highlight the effect of hypoxia and Foxn1 on the protein profile and functionality of regenerative Foxn1-/- DFs and demonstrate that the introduction of Foxn1 and Hif-1α into the wounded skin of regenerative Foxn1-/- mice activates reparative/scar-forming healing.
Keywords: Dermal fibroblasts; Foxn1; Hypoxia; Pai-1; Regeneration; Skin wound healing.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
Dermal White Adipose Tissue (dWAT) Is Regulated by Foxn1 and Hif-1α during the Early Phase of Skin Wound Healing.Int J Mol Sci. 2021 Dec 27;23(1):257. doi: 10.3390/ijms23010257. Int J Mol Sci. 2021. PMID: 35008683 Free PMC article.
-
Impairment of the Hif-1α regulatory pathway in Foxn1-deficient (Foxn1-/- ) mice affects the skin wound healing process.FASEB J. 2021 Feb;35(2):e21289. doi: 10.1096/fj.202001907R. FASEB J. 2021. PMID: 33475195 Free PMC article.
-
Effect of TGFβ1, TGFβ3 and keratinocyte conditioned media on functional characteristics of dermal fibroblasts derived from reparative (Balb/c) and regenerative (Foxn1 deficient; nude) mouse models.Cell Tissue Res. 2018 Oct;374(1):149-163. doi: 10.1007/s00441-018-2836-8. Epub 2018 Apr 10. Cell Tissue Res. 2018. PMID: 29637306 Free PMC article.
-
Foxn1 in Skin Development, Homeostasis and Wound Healing.Int J Mol Sci. 2018 Jul 4;19(7):1956. doi: 10.3390/ijms19071956. Int J Mol Sci. 2018. PMID: 29973508 Free PMC article. Review.
-
Animal models of skin regeneration.Reprod Biol. 2014 Mar;14(1):61-7. doi: 10.1016/j.repbio.2014.01.004. Epub 2014 Jan 17. Reprod Biol. 2014. PMID: 24607256 Review.
Cited by
-
Development and functional evaluation of recombinant type III collagen intrauterine implant gel.Regen Biomater. 2025 Mar 17;12:rbaf013. doi: 10.1093/rb/rbaf013. eCollection 2025. Regen Biomater. 2025. PMID: 40196171 Free PMC article.
-
Exploring the Potential of Extracellular Vesicles from Atlantic Cod (Gadus morhua L.) Serum and Mucus for Wound Healing In Vitro.Biology (Basel). 2025 Jul 17;14(7):870. doi: 10.3390/biology14070870. Biology (Basel). 2025. PMID: 40723426 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

