HBHA induces IL-10 from CD4+ T cells in patients with active tuberculosis but IFN-γ and IL-17 from individuals with Mycobacterium tuberculosis infection
- PMID: 39257584
- PMCID: PMC11384583
- DOI: 10.3389/fimmu.2024.1422700
HBHA induces IL-10 from CD4+ T cells in patients with active tuberculosis but IFN-γ and IL-17 from individuals with Mycobacterium tuberculosis infection
Abstract
Background: To effectively control tuberculosis (TB), it is crucial to distinguish between active TB disease and latent TB infection (LTBI) to provide appropriate treatment. However, no such tests are currently available. Immune responses associated with active TB and LTBI are dynamic and exhibit distinct patterns. Comparing these differences is crucial for developing new diagnostic methods and understanding the etiology of TB. This study aimed to investigate the relationship between pro- and anti-inflammatory CD4+ cytokine production following stimulation with two types of latency-associated Mycobacterium tuberculosis (M.tb) antigens to allow differentiation between active TB and LTBI.
Methods: Cryopreserved PBMCs from patients with active TB disease or LTBI were stimulated overnight with replication-related antigen [ESAT-6/CFP-10 (E/C)] or two latency-associated antigens [heparin-binding hemagglutinin (HBHA) and alpha-crystallin-like protein (Acr)]. Responses were analyzed using multiparameter flow cytometry: active TB disease (n=15), LTBI (n=15) and ELISA: active TB disease (n=26) or LTBI (n=27).
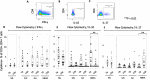
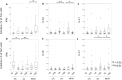
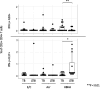
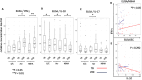
Results: CD4+ central memory T cells (Tcm) specific to E/C and CD4+ effector memory T cells specific to Acr and HBHA were higher in LTBI than in TB patients. IFN-γ+Tcm and IL-17+ Tem cells was higher in the LTBI group (p= 0.012 and p=0.029 respectively), but IL-10+ Tcm was higher in the active TB group (p= 0.029) following HBHA stimulation. Additionally, following stimulation with HBHA, IL-10 production from CD4+ T cells was significantly elevated in patients with active TB compared to those with LTBI (p= 0.0038), while CD4+ T cell production of IL-17 and IFN-γ was significantly elevated in LTBI compared to active TB (p= 0.0076, p< 0.0001, respectively). HBHA also induced more CCR6+IL-17+CD4Tcells and IL-17+FoxP3+CD25+CD4Tcells in LTBI than in TB patients (P=0.026 and P=0.04, respectively). HBHA also induced higher levels of IFN-γ+IL-10+CD4+ T cells in patients with active TB (Pp=0.03) and higher levels of IFN-γ+IL-17+ CD4+ T cells in those with LTBI (p=0.04). HBHA-specific cytokine production measured using ELISA showed higher levels of IFN-γ in participants with LTBI (P=0.004) and higher levels of IL-10 in those with active TB (P=0.04).
Conclusion: Stimulation with HBHA and measurement of CD4+ T cell production of IFN-γ, IL-10, and IL-17 could potentially differentiate active TB from LTBI. The characteristics of cytokine-expressing cells induced by HBHA also differed between participants with active TB and LTBI.
Keywords: HBHA; IFN-γ; IL-10; IL-17; LTBI; Mycobacterium tuberculosis (M.tb); TB.
Copyright © 2024 Izumida, Jobe, Coker, Barry, Rashid, Manneh, Daffeh, Ariyoshi and Sutherland.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures


References
-
- Fehily SR, Al-Ani AH, Abdelmalak J, Rentch C, Zhang E, Denholm JT, et al. Review article: latent tuberculosis in patients with inflammatory bowel diseases receiving immunosuppression-risks, screening, diagnosis and management. Aliment Pharmacol Ther. (2022) 56:6–27. doi: 10.1111/apt.16952 - DOI - PMC - PubMed
-
- World Health Organization . WHO consolidated guidelines on tuberculosis: tuberculosis preventive treatment Vol. 56. Genève, Switzerland: World Health Organization; (2020). p. - PubMed
-
- Zar HJ, Workman L, Isaacs W, Dheda K, Zemanay W, Nicol MP. Rapid diagnosis of pulmonary tuberculosis in African children in a primary care setting by use of Xpert MTB/RIF on respiratory specimens: a prospective study. Lancet Glob Health. (2013) 1:e97–e104. doi: 10.1016/S2214-109X(13)70036-6 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

