A Novel Engineering Cell Therapy Platform Mimicking the Immune Thrombocytopenia-Derived Platelets to Inhibit Cytokine Storm in Hemophagocytic Lymphohistiocytosis
- PMID: 39258712
- PMCID: PMC11615807
- DOI: 10.1002/advs.202404571
A Novel Engineering Cell Therapy Platform Mimicking the Immune Thrombocytopenia-Derived Platelets to Inhibit Cytokine Storm in Hemophagocytic Lymphohistiocytosis
Abstract
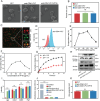
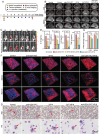
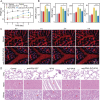
Hemophagocytic lymphohistiocytosis (HLH) is a common and highly fatal hyperinflammatory syndrome characterized by the aberrant activation of macrophages. To date, there is a lack of targeted therapies for HLH. It is validated that macrophages in HLH efficiently phagocytose anti-CD41-platelets (anti-CD41-PLTs) from immune thrombocytopenia (ITP) patients in previous research. Hence, the pathological mechanisms of ITP are mimicked and anti-CD41-PLTs are utilized to load the macrophage-toxic drug VP16 to construct macrophage-targetable engineered platelets anti-CD41-PLT-VP16, which is a novel targeted therapy against HLH. Both in vitro and in vivo studies demonstrate that anti-CD41-PLT-VP16 has excellent targeting and pro-macrophage apoptotic effects. In HLH model mice, anti-CD41-PLT-VP16 prevents hemophagocytosis and inhibits the cytokine storm. Mechanistic studies reveal that anti-CD41-PLT-VP16 increases the cytotoxicity of VP16, facilitating precise intervention in macrophages. Furthermore, it operates as a strategic "besieger" in diminishing hyperinflammation syndrome, which can indirectly prevent the abnormal activation of T cells and NK cells and reduce the Ab-dependent cell-mediated cytotoxicity effect. The first platelet-based clinical trial is ongoing. The results show that after treatment with anti-CD41-PLT-VP16, HLH patients have a threefold increase in the overall response rate compared to patients receiving conventional chemotherapy. In conclusion, anti-CD41-PLT-VP16 provides a general insight into hyperinflammation syndrome and offers a novel clinical therapeutic strategy for HLH.
Keywords: cytokine storm; etoposide; hemophagocytic lymphohistiocytosis; living therapeutics platform; platelets.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Cytokine Storm Syndrome.Annu Rev Med. 2023 Jan 27;74:321-337. doi: 10.1146/annurev-med-042921-112837. Epub 2022 Oct 13. Annu Rev Med. 2023. PMID: 36228171 Review.
-
The pathophysiology of hemophagocytic lympho-histiocytosis (HLH) syndrome and insights from animal models.Clin Exp Immunol. 2025 Jan 21;219(1):uxaf036. doi: 10.1093/cei/uxaf036. Clin Exp Immunol. 2025. PMID: 40435290 Review.
-
COVID-19-associated cytokine storm syndrome and diagnostic principles: an old and new Issue.Emerg Microbes Infect. 2021 Dec;10(1):266-276. doi: 10.1080/22221751.2021.1884503. Emerg Microbes Infect. 2021. PMID: 33522893 Free PMC article. Review.
-
Hemophagocytic lymphohistiocytosis: a review inspired by the COVID-19 pandemic.Rheumatol Int. 2021 Jan;41(1):7-18. doi: 10.1007/s00296-020-04636-y. Epub 2020 Jun 25. Rheumatol Int. 2021. PMID: 32588191 Free PMC article. Review.
-
Ruxolitinib-loaded cytokine nanosponge alleviated the cytokine storm and dampened macrophage overactivation for the treatment of hemophagocytic lymphohistiocytosis.Int J Pharm. 2024 May 25;657:124127. doi: 10.1016/j.ijpharm.2024.124127. Epub 2024 Apr 14. Int J Pharm. 2024. PMID: 38621611
References
-
- Hayden A., Park S., Giustini D., Lee A. Y. Y., Chen L. Y. C., Blood Rev. 2016, 30, 411. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
