Perioperative glucose monitoring with continuous glucose monitors identifies risk factors for post-transplant diabetes mellitus in kidney transplant recipients
- PMID: 39261619
- PMCID: PMC11390710
- DOI: 10.1038/s41598-024-72025-7
Perioperative glucose monitoring with continuous glucose monitors identifies risk factors for post-transplant diabetes mellitus in kidney transplant recipients
Abstract
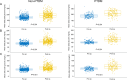
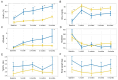
Post-transplantation diabetes mellitus (PTDM) negatively affects graft and patient survival after kidney transplantation (KT). This prospective study used continuous glucose monitoring (CGM) to evaluate perioperative blood glucose dynamics, identify PTDM risk factors, and compare predictive accuracy with capillary blood glucose monitoring (CBGM) in 60 non-diabetic living-donor KT recipients. Patients underwent 2-week pre- and postoperative CGM, including routine CBGM during their in-hospital stays. PTDM-related risk factors and glucose profiles were analyzed with postoperative CGM and CBG. PTDM developed in 14 (23.3%) patients and was associated with older age, male sex, higher baseline HbA1c, high-density lipoprotein cholesterol, and 3-month cumulative tacrolimus exposure levels. Male sex and postoperative time above the range (TAR) of 180 mg/dL by CGM were PTDM-related risk factors in the multivariate analysis. For predictive power, the CGM model with postoperative glucose profiles exhibited higher accuracy compared with the CBGM model (areas under the curves of 0.916, and 0.865, respectively). Therefore, we found that male patients with a higher postoperative TAR of 180 mg/dL have an increased risk of PTDM. Postoperative CGM provides detailed glucose dynamics and demonstrates superior predictive potential for PTDM than CBGM.
Keywords: Blood glucose; Continuous glucose monitoring; Diabetes mellitus; Follow up; Kidney transplantation; Postoperative care.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Continuous glucose monitoring for the prediction of posttransplant diabetes mellitus and impaired glucose tolerance on day 90 after kidney transplantation-A prospective proof-of-concept study.Am J Transplant. 2024 Dec;24(12):2225-2234. doi: 10.1016/j.ajt.2024.07.016. Epub 2024 Jul 22. Am J Transplant. 2024. PMID: 39047976
-
Oral Glucose Tolerance Test as a Risk Marker for Developing Post-Transplant Diabetes Mellitus.Transplant Proc. 2024 Jun;56(5):1061-1065. doi: 10.1016/j.transproceed.2024.04.021. Epub 2024 May 18. Transplant Proc. 2024. PMID: 38762406
-
Continuous glucose monitoring after kidney transplantation in non-diabetic patients: early hyperglycaemia is frequent and may herald post-transplantation diabetes mellitus and graft failure.Diabetes Metab. 2013 Oct;39(5):404-10. doi: 10.1016/j.diabet.2012.10.007. Epub 2013 Aug 30. Diabetes Metab. 2013. PMID: 23999231
-
An update review of post-transplant diabetes mellitus: Concept, risk factors, clinical implications and management.Diabetes Obes Metab. 2024 Jul;26(7):2531-2545. doi: 10.1111/dom.15575. Epub 2024 Apr 1. Diabetes Obes Metab. 2024. PMID: 38558257 Review.
-
Post-transplantation diabetes in kidney transplant recipients: an update on management and prevention.Acta Diabetol. 2018 Aug;55(8):763-779. doi: 10.1007/s00592-018-1137-8. Epub 2018 Apr 4. Acta Diabetol. 2018. PMID: 29619563 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

