Clinical Phenotyping for Prognosis and Immunotherapy Guidance in Bacterial Sepsis and COVID-19
- PMID: 39263383
- PMCID: PMC11390041
- DOI: 10.1097/CCE.0000000000001153
Clinical Phenotyping for Prognosis and Immunotherapy Guidance in Bacterial Sepsis and COVID-19
Abstract
Objectives: It is suggested that sepsis may be classified into four clinical phenotypes, using an algorithm employing 29 admission parameters. We applied a simplified phenotyping algorithm among patients with bacterial sepsis and severe COVID-19 and assessed characteristics and outcomes of the derived phenotypes.
Design: Retrospective analysis of data from prospective clinical studies.
Setting: Greek ICUs and Internal Medicine departments.
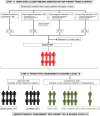
Patients and interventions: We analyzed 1498 patients, 620 with bacterial sepsis and 878 with severe COVID-19. We implemented a six-parameter algorithm (creatinine, lactate, aspartate transaminase, bilirubin, C-reactive protein, and international normalized ratio) to classify patients with bacterial sepsis intro previously defined phenotypes. Patients with severe COVID-19, included in two open-label immunotherapy trials were subsequently classified. Heterogeneity of treatment effect of anakinra was assessed. The primary outcome was 28-day mortality.
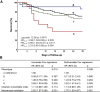
Measurements and main results: The algorithm validated the presence of the four phenotypes across the cohort of bacterial sepsis and the individual studies included in this cohort. Phenotype α represented younger patients with low risk of death, β was associated with high comorbidity burden, and δ with the highest mortality. Phenotype assignment was independently associated with outcome, even after adjustment for Charlson Comorbidity Index. Phenotype distribution and outcomes in severe COVID-19 followed a similar pattern.
Conclusions: A simplified algorithm successfully identified previously derived phenotypes of bacterial sepsis, which were predictive of outcome. This classification may apply to patients with severe COVID-19 with prognostic implications.
Keywords: COVID-19; immunotherapy; mortality; phenotypes; sepsis.
Copyright © 2024 The Authors. Published by Wolters Kluwer Health, Inc. on behalf of the Society of Critical Care Medicine.
Conflict of interest statement
Dr. Karakike has received funding by the Horizon2020 Marie Skłodowska-Curie International Training Network “the European Sepsis Academy” (grant number 676129- granted to the National and Kapodistrian University of Athens). Dr. Poulakou reports receiving grant funding and/or speaker’s honoraria from Gilead, Menarini, Merck Sharp & Dohme (MSD), Pfizer, Roche, and Sobi. Dr. Panagopoulos has received honoraria from GILEAD Sciences, Janssen, and MSD. Dr. Milionis reports receiving honoraria, consulting fees, and nonfinancial support from healthcare companies, including Amgen, Angelini, Bayer, Mylan, MSD, Pfizer, and Servier. Dr. Dalekos has acted as advisor/lecturer for Abbvie, Bristol-Myers Squibb, Gilead, Novartis, Roche, Amgen, MSD, Janssen, Ipsen, Genkyotex, Sobi, and Pfizer; he has received grant support from Bristol-Myers Squib, Gilead, Roche, Janssen, Abbvie, and Bayer; and he was or is currently principal investigator in national and international protocols sponsored by Abbvie, Bristol-Myers Squibb, Novartis, Gilead, Novo Nordisk, Genkyotex, Regulus Therapeutics, Tiziana Life Sciences, Bayer, Astellas, Ipsen, Pfizer, Amyndas Pharamaceuticals, CymaBay Therapeutics, and Roche. Dr. Giamarellos-Bourboulis has received honoraria from Abbott Products Operations, bioMérieux, Brahms GmbH, GlaxoSmithKline, InflaRx GmbH, Pfizer, and Swedish Orphan BioVitrum; he received independent educational grants from Abbott Products Operations, bioMérieux, Johnson & Johnson, MSD, Union Chimique Belge, and Swedish Orphan BioVitrum; and he received funding from the Horizon 2020 European grants ImmunoSep and Optimal use of hospital resources and intervention using suPAR for improving prognosis and care for patients with COVID-19 and the Horizon Health grant Equine Polyclonal antibodies Immunotherapy against COVID-19/SARS-CoV2–VOC (granted to the Hellenic Institute for the Study of Sepsis). The remaining authors have disclosed that they do not have any potential conflicts of interest.
Figures

References
-
- World Health Organization: Coronavirus disease (COVID-19) pandemic. Available at: https://www.who.int/emergencies/diseases/novel-coronavirus-2019. Accessed February 24, 2024.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

