Datopotamab Deruxtecan Versus Chemotherapy in Previously Treated Inoperable/Metastatic Hormone Receptor-Positive Human Epidermal Growth Factor Receptor 2-Negative Breast Cancer: Primary Results From TROPION-Breast01
- PMID: 39265124
- PMCID: PMC11771365
- DOI: 10.1200/JCO.24.00920
Datopotamab Deruxtecan Versus Chemotherapy in Previously Treated Inoperable/Metastatic Hormone Receptor-Positive Human Epidermal Growth Factor Receptor 2-Negative Breast Cancer: Primary Results From TROPION-Breast01
Abstract
Purpose: The global, phase 3, open-label, randomized TROPION-Breast01 study assessed the trophoblast cell surface antigen 2-directed antibody-drug conjugate datopotamab deruxtecan (Dato-DXd) versus investigator's choice of chemotherapy (ICC) in hormone receptor-positive/human epidermal growth factor receptor 2-negative (HR+/HER2-) breast cancer.
Methods: Adult patients with inoperable/metastatic HR+/HER2‒ breast cancer, who had disease progression on endocrine therapy, for whom endocrine therapy was unsuitable, and had received one to two previous lines of chemotherapy in the inoperable/metastatic setting, were randomly assigned 1:1 to Dato-DXd (6 mg/kg once every 3 weeks) or ICC (eribulin/vinorelbine/capecitabine/gemcitabine). Dual primary end points were progression-free survival (PFS) by blinded independent central review (BICR) and overall survival (OS).
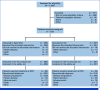
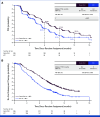
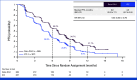
Results: Patients were randomly assigned to Dato-DXd (n = 365) or ICC (n = 367). Dato-DXd significantly reduced the risk of progression or death versus ICC (PFS by BICR hazard ratio [HR], 0.63 [95% CI, 0.52 to 0.76]; P < .0001). Consistent PFS benefit was observed across subgroups. Although OS data were not mature, a trend favoring Dato-DXd was observed (HR, 0.84 [95% CI, 0.62 to 1.14]). The rate of grade ≥3 treatment-related adverse events (TRAEs) with Dato-DXd was lower than ICC (20.8% v 44.7%). The most common TRAEs (any grade; grade ≥3) were nausea (51.1%; 1.4%) and stomatitis (50%; 6.4%) with Dato-DXd and neutropenia (grouped term, 42.5%; 30.8%) with ICC.
Conclusion: Patients receiving Dato-DXd had statistically significant and clinically meaningful improvement in PFS and a favorable and manageable safety profile, compared with ICC. Results support Dato-DXd as a novel treatment option for patients with inoperable/metastatic HR+/HER2‒ breast cancer who have received one to two previous lines of chemotherapy in this setting.
Trial registration: ClinicalTrials.gov NCT05104866.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
Figures

References
-
- Gennari A, André F, Barrios CH, et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann Oncol. 2021;32:1475–1495. - PubMed
-
- Jhaveri K, Marmé F. Current and emerging treatment approaches for hormone receptor-positive/human epidermal growth factor receptor 2-negative metastatic breast cancer. Cancer Treat Rev. 2024;123:102670. - PubMed
-
- Cortes J, O'Shaughnessy J, Loesch D, et al. Eribulin monotherapy versus treatment of physician's choice in patients with metastatic breast cancer (EMBRACE): A phase 3 open-label randomised study. Lancet. 2011;377:914–923. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

