Phosphorylated Tau in the Taste Buds of Alzheimer's Disease Mouse Models
- PMID: 39266476
- PMCID: PMC11411091
- DOI: 10.5607/en24004
Phosphorylated Tau in the Taste Buds of Alzheimer's Disease Mouse Models
Abstract
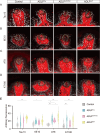
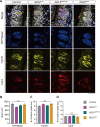
Numerous systemic diseases manifest with oral symptoms and signs. The molecular diagnosis of Alzheimer's disease (AD), the most prevalent neurodegenerative disease worldwide, currently relies on invasive or expensive methods, emphasizing the imperative for easily accessible biomarkers. In this study, we explored the expression patterns of key proteins implicated in AD pathophysiology within the taste buds of mice. We detected the expression of amyloid precursor protein (APP) and tau protein in the taste buds of normal C57BL/6 mice. Phosphorylated tau was predominantly found in type II and III taste cells, while APP was located in type I taste cells. Remarkably, we observed significantly stronger immunoreactivity to phosphorylated tau in the taste buds of aged AD mouse models compared to age-matched controls. These findings underscore the oral expression of biomarkers associated with AD, highlighting the diagnostic potential of the oral cavity for neurodegenerative diseases.
Keywords: Alzheimer’s disease; Amyloid protein precursor; Immunohistochemistry; Taste buds; Tauopathies.
Figures



References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

