Safety, Tolerability, and Short-Term Efficacy of Low-Level Light Therapy for Dry Age-Related Macular Degeneration
- PMID: 39271642
- PMCID: PMC11494651
- DOI: 10.1007/s40123-024-01030-w
Safety, Tolerability, and Short-Term Efficacy of Low-Level Light Therapy for Dry Age-Related Macular Degeneration
Abstract
Introduction: Photobiomodulation (PBM) has become a promising approach for slowing the progression of early and intermediate dry age-related macular degeneration (dAMD) to advanced AMD. This technique uses light to penetrate tissues and activate molecules that influence biochemical reactions and cellular metabolism. This preliminary analysis is aimed at assessing the safety, tolerability, and short-term effectiveness of the EYE-LIGHT®PBM treatment device in patients with dAMD.
Methods: The EYE-LIGHT® device employs two wavelengths, 590 nm (yellow) and 630 nm (red), in both continuous and pulsed modes. Patients over 50 years of age with a diagnosis of dAMD in any AREDS (Age-Related Eye Disease Study) category were randomly assigned to either the treatment group or the sham group. The treatment plan consisted of an initial cycle of two sessions per week for 4 weeks. Safety, tolerability, and compliance outcomes, along with functional and anatomical outcomes, were assessed at the end of the fourth month.
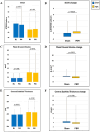
Results: This preliminary analysis included data from 76 patients (152 eyes). All patients were fully compliant with treatment sessions, and only one fifth of patients treated with PBM reported mild ocular adverse events, highlighting exceptional results in terms of tolerability and adherence. Changes in best-corrected visual acuity (BCVA) from baseline to month 4 differed significantly between the sham and PBM-treated groups, favoring the latter, with a higher proportion achieving a gain of five or more letters post-treatment (8.9% vs. 20.3%, respectively; p = 0.043). No significant differences in central subfield thickness (CST) were observed between the two groups over the 4-month period. The study also found a statistically significant disparity in mean drusen volume changes from baseline to month 4 between the groups in favor of patients treated with PBM (p = 0.013).
Conclusion: These preliminary results indicate that PBM treatment using the EYE-LIGHT® system is safe and well tolerated among patients with dAMD. Furthermore, both functional and anatomical data support the treatment's short-term efficacy.
Trial registration: ClinicalTrials.gov identifier NCT06046118.
Keywords: Age related macular degeneration; Drusen; Dry AMD; Low-level light therapy; Photobiomodulation.
© 2024. The Author(s).
Conflict of interest statement
Enrico Borrelli, Giulia Coco, Marco Pellegrini, Marco Mura, Nicolò Ciarmatori, Vincenzo Scorcia, Adriano Carnevali, Andrea Lucisano, Massimiliano Borselli, Costanza Rossi, Michele Reibaldi, Federico Ricardi, Aldo Vagge, Massimo Nicolò, Paolo Forte, Antonio Cartabellotta, Murat Hasanreisoğlu, Cem Kesim, Sibel Demirel, Özge Yanık, Federico Bernabei, Pierre-Raphael Rothschild, Sarah Farrant, and Giuseppe Giannaccare have no conflicts of interest to disclose.
Figures


References
-
- Fleckenstein M, Schmitz-Valckenberg S, Chakravarthy U. Age-related macular degeneration. JAMA. 2024;331:147. - PubMed
-
- Li JQ, Welchowski T, Schmid M, Mauschitz MM, Holz FG, Finger RP. Prevalence and incidence of age-related macular degeneration in Europe: a systematic review and meta-analysis. Br J Ophthalmol. 2020;104:1077–84. - PubMed
-
- Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration. JAMA. 2013;309:2005. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical

